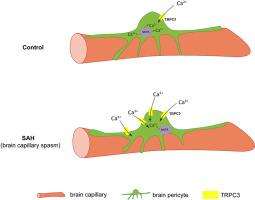
蛛网膜下腔出血后,TRPC3介导的钙离子流入脑周皮细胞的途径发生改变,导致毛细血管痉挛。
IF 5.6
2区 医学
Q1 CLINICAL NEUROLOGY
引用次数: 0
摘要
蛛网膜下腔出血后,钙离子的流入和随后细胞内钙浓度([Ca2+]i)的升高会引起脑周细胞收缩和毛细血管痉挛。钙的流入是通过阳离子通道实现的。然而,蛛网膜下腔出血后脑周细胞中特定的钙离子通道仍然未知。瞬时受体电位Canonical 3(TRPC3)是可能参与脑周质钙离子流入的最丰富的阳离子通道,它通过储存操作钙离子进入(SOCE)或受体操作钙离子进入(ROCE)参与其他类型细胞的钙离子流入。因此,我们假设 TRPC3 与脑周质中[Ca2+]i 的升高有关,有可能介导蛛网膜下腔出血后脑周质收缩和毛细血管痉挛。在这项研究中,我们分离了大鼠脑周细胞,并证实了蛛网膜下腔出血后脑周细胞中 TRPC3 表达及其电流的增加。脑周细胞的钙成像显示,TRPC3表达的变化介导了蛛网膜下腔出血后钙离子流入从SOCE主导型向ROCE主导型的转变,导致SAH后[Ca2+]i水平显著升高。在体内大鼠蛛网膜下腔出血模型中,脑周细胞中的 TRPC3 活性也导致了毛细血管痉挛和脑血流量减少。因此,我们认为 TRPC3 介导的钙离子流入途径的转换在蛛网膜下腔出血后脑周细胞[Ca2+]i 升高,最终导致毛细血管痉挛和脑血流减少中起着至关重要的作用。本文章由计算机程序翻译,如有差异,请以英文原文为准。
A switch in the pathway of TRPC3-mediated calcium influx into brain pericytes contributes to capillary spasms after subarachnoid hemorrhage
Calcium influx and subsequent elevation of the intracellular calcium concentration ([Ca2+]i) induce contractions of brain pericytes and capillary spasms following subarachnoid hemorrhage. This calcium influx is exerted through cation channels. However, the specific calcium influx pathways in brain pericytes after subarachnoid hemorrhage remain unknown. Transient receptor potential canonical 3 (TRPC3) is the most abundant cation channel potentially involved in calcium influx into brain pericytes and is involved in calcium influx into other cell types either via store-operated calcium entry (SOCE) or receptor-operated calcium entry (ROCE). Therefore, we hypothesized that TRPC3 is associated with [Ca2+]i elevation in brain pericytes, potentially mediating brain pericyte contraction and capillary spasms after subarachnoid hemorrhage. In this study, we isolated rat brain pericytes and demonstrated increased TRPC3 expression and its currents in brain pericytes after subarachnoid hemorrhage. Calcium imaging of brain pericytes revealed that changes in TRPC3 expression mediated a switch from SOCE-dominant to ROCE-dominant calcium influx after subarachnoid hemorrhage, resulting in significantly higher [Ca2+]i levels after SAH. TRPC3 activity in brain pericytes also contributed to capillary spasms and reduction in cerebral blood flow in an in vivo rat model of subarachnoid hemorrhage. Therefore, we suggest that the switch in TRPC3-mediated calcium influx pathways plays a crucial role in the [Ca2+]i elevation in brain pericytes after subarachnoid hemorrhage, ultimately leading to capillary spasms and a reduction in cerebral blood flow.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Neurotherapeutics
医学-神经科学
CiteScore
11.00
自引率
3.50%
发文量
154
审稿时长
6-12 weeks
期刊介绍:
Neurotherapeutics® is the journal of the American Society for Experimental Neurotherapeutics (ASENT). Each issue provides critical reviews of an important topic relating to the treatment of neurological disorders written by international authorities.
The Journal also publishes original research articles in translational neuroscience including descriptions of cutting edge therapies that cross disciplinary lines and represent important contributions to neurotherapeutics for medical practitioners and other researchers in the field.
Neurotherapeutics ® delivers a multidisciplinary perspective on the frontiers of translational neuroscience, provides perspectives on current research and practice, and covers social and ethical as well as scientific issues.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


