通过基于构件的分子网络方法,有针对性地从 Cleistocalyx operculatus 中分离出抗病毒的肉桂酰氯葡萄糖醇-萜烯加合物
IF 14.7
1区 医学
Q1 PHARMACOLOGY & PHARMACY
引用次数: 0
摘要
通过广泛的光谱方法、量子化学计算和单晶 X 射线晶体学实验,确定了它们的结构(包括绝对构型)。Cleistoperone A()由一个肉桂酰氯葡萄糖醇基团和两个线性单萜分子组成,是一种前所未有的大环 CPTA,其密集官能化的三环[15.3.1.0]庚桥环骨架包含一个可烯醇化的′-三酮系统和两种不同的立体元素(包括五个点手性和三个平面手性)。Cleistoperones B 和 C(和)是两种新的 CPTA,其(nor)单萜烯分子与肉桂酰氯葡萄糖醇单元的肉桂酰链之间具有不同寻常的偶联模式。Cleistoperone D()具有前所未有的笼状 6/6/6/4/6 融合杂五环支架。此外,还提出了 - 的合理生物合成途径。值得注意的是,、、、和化合物对呼吸道合胞病毒(RSV)具有显著的抗病毒活性。其中药效最强的是cleistoperone A(),其IC值为1.71 ± 0.61 μmol/L,可通过影响Akt/mTOR/p70S6K信号通路有效抑制病毒复制。本文章由计算机程序翻译,如有差异,请以英文原文为准。
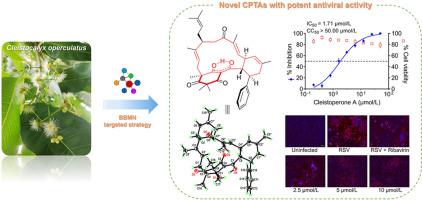
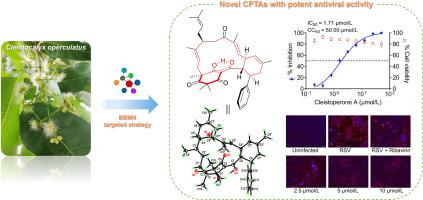
Targeted isolation of antiviral cinnamoylphloroglucinol-terpene adducts from Cleistocalyx operculatus by building blocks-based molecular networking approach
The building blocks-based molecular network (BBMN) strategy was applied to the phytochemical investigation of Cleistocalyx operculatus, leading to the targeted isolation of eighteen novel cinnamoylphloroglucinol-terpene adducts (CPTAs) with diverse skeleton types (cleistoperones A–R, 1–18). Their structures including absolute configurations were determined by extensive spectroscopic methods, quantum chemical calculations, and single-crystal X-ray crystallographic experiments. Cleistoperone A (1), consisting of a cinnamoylphloroglucinol motif and two linear monoterpene moieties, represents an unprecedented macrocyclic CPTA, whose densely functionalized tricyclo[15.3.1.03,8]heneicosane bridge ring skeleton contains an enolizable β,β′-triketone system and two different kinds of stereogenic elements (including five point and three planar chiralities). Cleistoperones B and C (2 and 3) are two new skeletal CPTAs with an unusual coupling pattern between the (nor)monoterpene moiety and the cinnamoyl chain of the cinnamoylphloroglucinol unit. Cleistoperone D (4) possesses an unprecedented cage-like 6/6/6/4/6-fused heteropentacyclic scaffold. The plausible biosynthetic pathways for 1–18 were also proposed. Notably, compounds 1, 4, 7, 8, and 18 exhibited significant antiviral activity against respiratory syncytial virus (RSV). The most potent one, cleistoperone A (1) with IC50 value of 1.71 ± 0.61 μmol/L, could effectively inhibit virus replication via affecting the Akt/mTOR/p70S6K signaling pathway.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Acta Pharmaceutica Sinica. B
Pharmacology, Toxicology and Pharmaceutics-General Pharmacology, Toxicology and Pharmaceutics
CiteScore
22.40
自引率
5.50%
发文量
1051
审稿时长
19 weeks
期刊介绍:
The Journal of the Institute of Materia Medica, Chinese Academy of Medical Sciences, and the Chinese Pharmaceutical Association oversees the peer review process for Acta Pharmaceutica Sinica. B (APSB).
Published monthly in English, APSB is dedicated to disseminating significant original research articles, rapid communications, and high-quality reviews that highlight recent advances across various pharmaceutical sciences domains. These encompass pharmacology, pharmaceutics, medicinal chemistry, natural products, pharmacognosy, pharmaceutical analysis, and pharmacokinetics.
A part of the Acta Pharmaceutica Sinica series, established in 1953 and indexed in prominent databases like Chemical Abstracts, Index Medicus, SciFinder Scholar, Biological Abstracts, International Pharmaceutical Abstracts, Cambridge Scientific Abstracts, and Current Bibliography on Science and Technology, APSB is sponsored by the Institute of Materia Medica, Chinese Academy of Medical Sciences, and the Chinese Pharmaceutical Association. Its production and hosting are facilitated by Elsevier B.V. This collaborative effort ensures APSB's commitment to delivering valuable contributions to the pharmaceutical sciences community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


