在大型动物模型中,视网膜下 AAV 释放靶向 VEGFA 的 RNAi- 治疗药物可减少脉络膜新生血管的形成
IF 4.7
2区 医学
Q2 MEDICINE, RESEARCH & EXPERIMENTAL
Molecular Therapy-Methods & Clinical Development
Pub Date : 2024-03-22
DOI:10.1016/j.omtm.2024.101242
引用次数: 0
摘要
新生血管性老年黄斑变性(nAMD)是西方国家老年人视力丧失的常见原因。目前通过反复注射抗血管内皮生长因子药物来治疗疾病的方法会累积不良反应风险,给社会和患者个人造成负担。在猪脉络膜新生血管(CNV)模型中,我们利用基于腺相关病毒(AAV)的新型 RNA 干扰(RNAi)效应物进行了探讨。在小鼠中建立并验证了置于 pri-microRNA 支架(miR-agshRNA)中的 Ago2 依赖性短发夹 RNA 的靶向效力。随后,在激光诱导 CNV 之前,通过视网膜下注射将编码-靶向或无关 miR-agshRNA 的 AAV 血清型 8(AAV2.8)载体送入猪视网膜。值得注意的是,与非靶向对照组相比,靶向 miR-agshRNAs 能显著减少 CNV。我们还证明了单链和自补体 AAV2.8 载体能有效转导猪视网膜色素上皮细胞,但它们的转导特性和视网膜安全性不同。总之,我们的数据表明,在一个大型转化动物模型中,靶向 miR-aghsRNAs 具有强大的抗血管生成作用,从而表明基于 AAV 的抗 VEGFA RNAi 治疗药物是治疗 nAMD 的一种有价值的工具。本文章由计算机程序翻译,如有差异,请以英文原文为准。
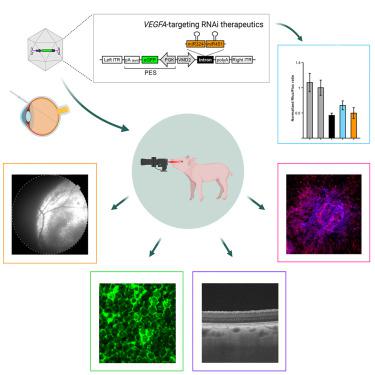
Subretinal AAV delivery of RNAi-therapeutics targeting VEGFA reduces choroidal neovascularization in a large animal model
Neovascular age-related macular degeneration (nAMD) is a frequent cause of vision loss among the elderly in the Western world. Current disease management with repeated injections of anti-VEGF agents accumulates the risk for adverse events and constitutes a burden for society and the individual patient. Sustained suppression of VEGF using gene therapy is an attractive alternative, which we explored using adeno-associated virus (AAV)-based delivery of novel RNA interference (RNAi) effectors in a porcine model of choroidal neovascularization (CNV). The potency of -targeting, Ago2-dependent short hairpin RNAs placed in pri-microRNA scaffolds (miR-agshRNA) was established and in mice. Subsequently, AAV serotype 8 (AAV2.8) vectors encoding -targeting or irrelevant miR-agshRNAs under the control of a tissue-specific promotor were delivered to the porcine retina via subretinal injection before CNV induction by laser. Notably, -targeting miR-agshRNAs resulted in a significant and sizable reduction of CNV compared with the non-targeting control. We also demonstrated that single-stranded and self-complementary AAV2.8 vectors efficiently transduce porcine retinal pigment epithelium cells but differ in their transduction characteristics and retinal safety. Collectively, our data demonstrated a robust anti-angiogenic effect of -targeting miR-aghsRNAs in a large translational animal model, thereby suggesting AAV-based delivery of anti-VEGFA RNAi therapeutics as a valuable tool for the management of nAMD.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Molecular Therapy-Methods & Clinical Development
Biochemistry, Genetics and Molecular Biology-Molecular Biology
CiteScore
9.90
自引率
4.30%
发文量
163
审稿时长
12 weeks
期刊介绍:
The aim of Molecular Therapy—Methods & Clinical Development is to build upon the success of Molecular Therapy in publishing important peer-reviewed methods and procedures, as well as translational advances in the broad array of fields under the molecular therapy umbrella.
Topics of particular interest within the journal''s scope include:
Gene vector engineering and production,
Methods for targeted genome editing and engineering,
Methods and technology development for cell reprogramming and directed differentiation of pluripotent cells,
Methods for gene and cell vector delivery,
Development of biomaterials and nanoparticles for applications in gene and cell therapy and regenerative medicine,
Analysis of gene and cell vector biodistribution and tracking,
Pharmacology/toxicology studies of new and next-generation vectors,
Methods for cell isolation, engineering, culture, expansion, and transplantation,
Cell processing, storage, and banking for therapeutic application,
Preclinical and QC/QA assay development,
Translational and clinical scale-up and Good Manufacturing procedures and process development,
Clinical protocol development,
Computational and bioinformatic methods for analysis, modeling, or visualization of biological data,
Negotiating the regulatory approval process and obtaining such approval for clinical trials.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


