基因治疗产品加速计划的分析和比较评估:美国、欧盟、日本和韩国的见解
IF 4.6
3区 医学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
摘要
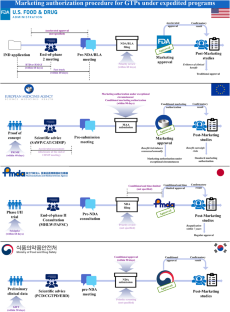
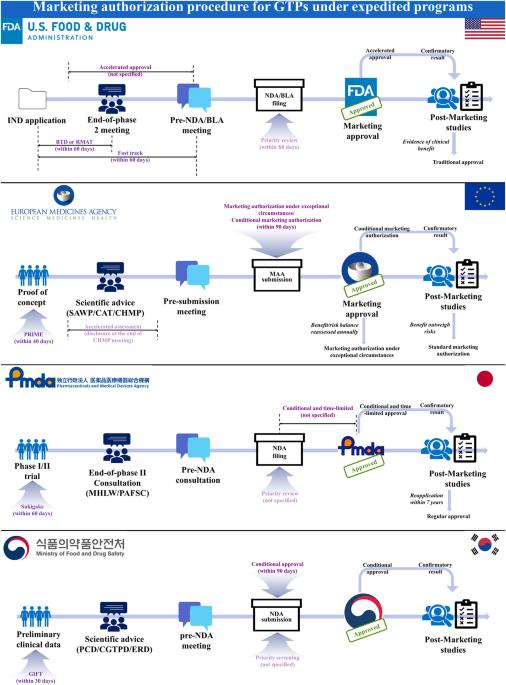
用于治疗不治之症的基因治疗产品(GTP)可以加快早期商业化进程,以满足尚未满足的需求。本研究利用美国、欧盟、日本和韩国监管机构的网站、相关法规和文件,对这些国家的基因治疗产品加速计划进行了分析。美国共有五项全球贸易点加速计划,欧盟有四项,日本和韩国各有三项,其中四项是为全球贸易点量身定制的。这些计划的目标相似,可归类为加快药物开发、审评和审批的计划。不过,各监管机构在资格标准、具体福利和上市后研究条件方面存在差异。此外,罕见病孤儿药的指定标准在患病率阈值、提供的激励措施和市场独占期方面也有所不同。总体而言,共有 19 种 GTP 获得批准--13 种在美国,14 种在欧盟,8 种在日本,3 种在韩国--其中大多数通过至少一种快速方案获得监管批准。因此,未来的研究可以分析,与只处理一个快速程序相比,获得多个快速程序是否会加速 GTPs 的药物开发和商业化。此外,我们还鼓励机构间进行科学讨论,以协调快速项目的要求。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Analysis and comparative evaluation of expedited programs for gene therapy products: insights from the United States, the European Union, Japan, and South Korea
Gene therapy products (GTPs) used for incurable diseases can be expedited for early commercialization to fulfill unmet needs. This study analyzed the expedited programs available for GTPs in the US, EU, Japan, and South Korea using their regulatory authorities’ websites, related regulations, and documents. In total, there were five expedited programs available for GTPs in the US, four in the EU, and three in both Japan and South Korea, of which four are tailored for GTPs. These programs, sharing similar objectives, can be categorized as those expediting drug development, review, and approval. However, variations are observed in eligibility criteria, specific benefits, and post-marketing study conditions across regulatory authorities. Additionally, the criteria for orphan drug designation for a rare disease differs in prevalence thresholds, incentive offered, and marketing exclusivity period. Overall, 19 GTPs were approved–13 in the US, 14 in the EU, eight in Japan, and three in South Korea–with majority obtaining regulatory approval through at least one expedited program. Therefore, future studies can analyze whether acquiring multiple expedited programs accelerates the drug development and commercialization of GTPs compared with when only one expedited program is processed. Additionally, inter-authority scientific discussion is encouraged for harmonization of expedited program requirements.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Gene Therapy
医学-生化与分子生物学
CiteScore
9.70
自引率
2.00%
发文量
67
审稿时长
4-8 weeks
期刊介绍:
Gene Therapy covers both the research and clinical applications of novel therapeutic techniques based on a genetic component. Over the last few decades, significant advances in technologies ranging from identifying novel genetic targets that cause disease through to clinical studies, which show therapeutic benefit, have elevated this multidisciplinary field to the forefront of modern medicine.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


