1,3-取代吡唑类亚甲基衍生物的合成、表征、分子对接研究及抗菌活性研究
IF 0.8
4区 化学
Q4 CHEMISTRY, ORGANIC
引用次数: 0
摘要
以3-(4-氯苯基)-1-苯基- 1h -吡唑-4-醛(3a)和3-(呋喃-2-基)-1-苯基- 1h -吡唑-4-醛(3b)为中间体,通过不同取代苯胺的缩合反应,合成了两个不同系列的1,3取代吡唑的亚甲基衍生物。中间体由相应的苯腙通过Vilsmeier-Haack反应得到。合成的化合物通过IR、1H NMR和质谱进行了表征。分子对接研究结果显示,化合物4a7和4a8对真菌分泌的靶天冬氨酸蛋白酶(Sap) 1 (2QZW)具有最高的结合亲和力,化合物4a8和4a5对细菌靶葡萄糖胺-6-磷酸合成酶(2VF5)具有最高的结合亲和力。测定相应的亚甲胺衍生物对革兰氏阳性菌和革兰氏阴性菌的体外抑菌活性。评估对白色念珠菌的抗真菌活性。结果表明,筛选的15个化合物中有6个具有良好的抗菌活性。其中4a1、4a4、4a6和4a7对白色念珠菌有较好的抑制作用。本文章由计算机程序翻译,如有差异,请以英文原文为准。
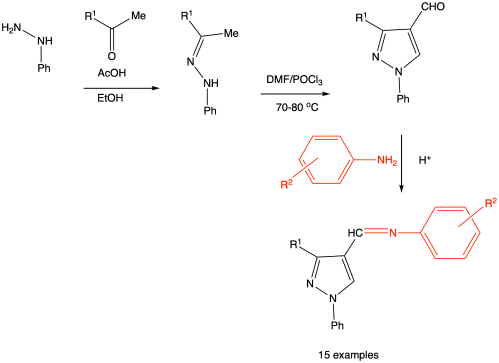
Synthesis, Characterization, Molecular Docking Studies and Antimicrobial Activity of Azomethine Derivatives of 1,3-Substituted Pyrazoles
Two different series of azomethine derivatives of 1,3-substitued pyrazoles were synthesized from the intermediates 3-(4-chlorophenyl)-1-phenyl-1H-pyrazole-4-carbaldehyde (3a) and 3-(furan-2-yl)-1-phenyl-1H-pyrazole-4-carbaldehyde (3b) by the condensation of various substituted anilines. The intermediates were obtained from the appropriate phenylhydrazones via Vilsmeier–Haack reaction. The synthesized compounds were characterized by IR, 1H NMR and MASS spectral studies. Molecular docking studies results revealed that 4a7 and 4a8 showed highest binding affinity against fungal target secreted aspartic proteinase (Sap) 1 (2QZW) and compounds 4a8 and 4a5 showed highest binding affinity against bacterial target glucosamine-6-phosphate synthase (2VF5). In vitro antimicrobial activity of corresponding azomethine derivatives were assessed on Gram-positive and Gram-negative bacteria. Antifungal activity was assessed on Candida albicans. The results revealed that six out of fifteen compounds screened have shown good antimicrobial activity. Among them, 4a1, 4a4, 4a6 and 4a7 shown good inhibition against Candida albicans.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Heterocycles
化学-有机化学
CiteScore
1.50
自引率
0.00%
发文量
108
审稿时长
1 months
期刊介绍:
Since its inception in 1973 HETEROCYCLES has provided a platform for the rapid exchange of research in the areas of organic, pharmaceutical, analytical, and medicinal chemistry of heterocyclic compounds in addition to communications, papers, reviews, a special section of the journal presents newly-discovered natural products whose structure has recently been established.
Another section is devoted to the total synthesis of previously documented natural products with heterocyclic ring systems.
Due to the fact that the journal is able to publish articles within two months of receipt of the manuscripts, researchers in this field can obtain up-to-date information on heterocyclic research by reading Heterocycles regularly.
Audience: Organic and Physical Organic Chemists, Biochemists, Pharmacologists and Scientists studying heterocyclic compounds
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


