三聚体线圈中b-, c-和f-残基之间的稳定协同作用的静电起源
IF 1.5
4区 生物学
Q4 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
摘要
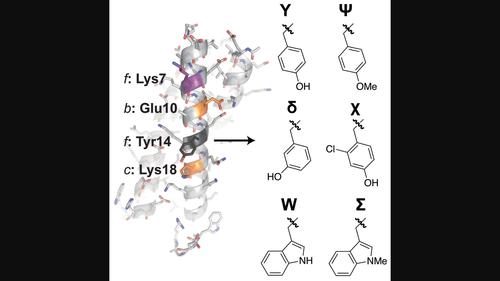
螺旋结构是最常见的蛋白质四级结构之一,是氨基酸序列和蛋白质构象之间最容易理解的关系。然而,残基在标准七位(a、d、e和g)上的作用被精确地详细地理解,传统的方法通常假设溶剂暴露的b、c和f位可以广泛地变化,以达到特定应用的目的,而后果最小。然而,越来越多的证据表明,这些b, c和f残基之间的相互作用可以在很大程度上有助于线圈的构象稳定性。在这里描述的三聚体线圈中,我们发现b位Glu10与c位Lys18进行稳定的远程协同作用(ΔΔΔGf =−0.65±0.02 kcal/mol)。这种有利的相互作用很大程度上取决于附近两个f位残基的存在:lys7和Tyr14。对这些残基在添加盐和变性剂存在下的广泛突变分析表明,这种远程协同作用主要是静电产生的,但也取决于f位Tyr14内侧链氢键供体的精确位置和酸度。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Electrostatic origin of a stabilizing synergistic interaction among b-, c-, and f-residues in a trimeric coiled coil
Coiled coils are one of most common protein quaternary structures and represent the best understood relationship between amino acid sequence and protein conformation. Whereas the roles of residues at the canonical heptad positions the a, d, e, and g are understood in precise detail, conventional approaches often assume that the solvent-exposed b-, c-, and f-positions can be varied broadly for application-specific purposes with minimal consequences. However, a growing body of evidence suggests that interactions among these b, c, and f residues can contribute substantially to coiled-coil conformational stability. In the trimeric coiled coil described here, we find that b-position Glu10 engages in a stabilizing long-range synergistic interaction with c-position Lys18 (ΔΔΔGf = −0.65 ± 0.02 kcal/mol). This favorable interaction depends strongly on the presence of two nearby f-position residues: Lys 7 and Tyr14. Extensive mutational analysis of these residues in the presence of added salt versus denaturant suggests that this long-range synergistic interaction is primarily electrostatic in origin, but also depends on the precise location and acidity of a side-chain hydrogen-bond donor within f-position Tyr14.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Peptide Science
Biochemistry, Genetics and Molecular Biology-Biophysics
CiteScore
5.20
自引率
4.20%
发文量
36
期刊介绍:
The aim of Peptide Science is to publish significant original research papers and up-to-date reviews covering the entire field of peptide research. Peptide Science provides a forum for papers exploring all aspects of peptide synthesis, materials, structure and bioactivity, including the use of peptides in exploring protein functions and protein-protein interactions. By incorporating both experimental and theoretical studies across the whole spectrum of peptide science, the journal serves the interdisciplinary biochemical, biomaterials, biophysical and biomedical research communities.
Peptide Science is the official journal of the American Peptide Society.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


