Bacterial structural genomics target enabled by a recently discovered potent fungal acetyl-CoA synthetase inhibitor
IF 1.1
4区 生物学
Q4 BIOCHEMICAL RESEARCH METHODS
Acta crystallographica. Section F, Structural biology communications
Pub Date : 2023-05-25
DOI:10.1107/S2053230X23003801
引用次数: 0
Abstract
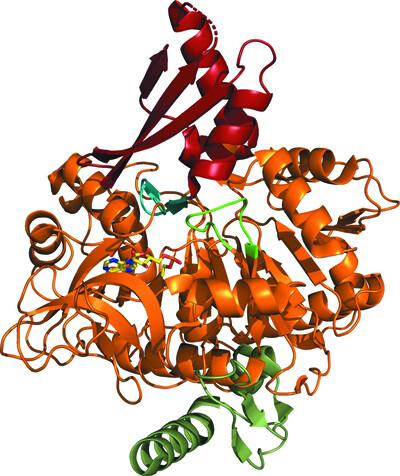
The compound ethyl-adenosyl monophosphate ester (ethyl-AMP) has been shown to effectively inhibit acetyl-CoA synthetase (ACS) enzymes and to facilitate the crystallization of fungal ACS enzymes in various contexts. In this study, the addition of ethyl-AMP to a bacterial ACS from Legionella pneumophila resulted in the determination of a co-crystal structure of this previously elusive structural genomics target. The dual functionality of ethyl-AMP in both inhibiting ACS enzymes and promoting crystallization establishes its significance as a valuable resource for advancing structural investigations of this class of proteins.
细菌结构基因组学目标启用了最近发现的有效真菌乙酰辅酶a合成酶抑制剂
化合物乙基腺苷单磷酸酯(乙基- amp)已被证明能有效抑制乙酰辅酶a合成酶(ACS)酶,并促进真菌ACS酶在各种环境下的结晶。在这项研究中,将乙基amp添加到嗜肺军团菌的细菌ACS中,从而确定了这种先前难以捉摸的结构基因组学靶点的共晶结构。乙基amp具有抑制ACS酶和促进结晶的双重功能,这为推进这类蛋白质的结构研究提供了宝贵的资源。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Acta crystallographica. Section F, Structural biology communications
BIOCHEMICAL RESEARCH METHODSBIOCHEMISTRY &-BIOCHEMISTRY & MOLECULAR BIOLOGY
CiteScore
1.90
自引率
0.00%
发文量
95
期刊介绍:
Acta Crystallographica Section F is a rapid structural biology communications journal.
Articles on any aspect of structural biology, including structures determined using high-throughput methods or from iterative studies such as those used in the pharmaceutical industry, are welcomed by the journal.
The journal offers the option of open access, and all communications benefit from unlimited free use of colour illustrations and no page charges. Authors are encouraged to submit multimedia content for publication with their articles.
Acta Cryst. F has a dedicated online tool called publBio that is designed to make the preparation and submission of articles easier for authors.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


