N6-methyladenosine-modified USP13 induces pro-survival autophagy and imatinib resistance via regulating the stabilization of autophagy-related protein 5 in gastrointestinal stromal tumors
IF 15.4
1区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 3
Abstract
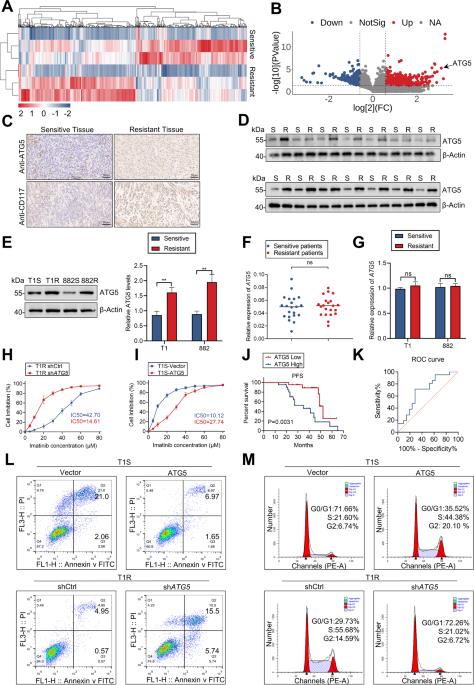
Secondary resistance to imatinib (IM) represents a major challenge for therapy of gastrointestinal stromal tumors (GISTs). Aberrations in oncogenic pathways, including autophagy, correlate with IM resistance. Regulation of autophagy-related protein 5 (ATG5) by the ubiquitin-proteasome system is critical for autophagic activity, although the molecular mechanisms that underpin reversible deubiquitination of ATG5 have not been deciphered fully. Here, we identified USP13 as an essential deubiquitinase that stabilizes ATG5 in a process that depends on the PAK1 serine/threonine-protein kinase and which enhances autophagy and promotes IM resistance in GIST cells. USP13 preferentially is induced in GIST cells by IM and interacts with ATG5, which leads to stabilization of ATG5 through deubiquitination. Activation of PAK1 promoted phosphorylation of ATG5 thereby enhancing the interaction of ATG5 with USP13. Furthermore, N6-methyladenosine methyltransferase-like 3 (METTL3) mediated stabilization of USP13 mRNA that required the m6A reader IGF2BP2. Moreover, an inhibitor of USP13 caused ATG5 decay and co-administration of this inhibitor with 3-methyladenine boosted treatment efficacy of IM in murine xenograft models derived from GIST cells. Our findings highlight USP13 as an essential regulator of autophagy and IM resistance in GIST cells and reveal USP13 as a novel potential therapeutic target for GIST treatment.
N6-甲基腺苷修饰的USP13通过调节胃肠道间质瘤中自噬相关蛋白5的稳定性诱导有利于生存的自噬和伊马替尼耐药性
伊马替尼(IM)的继发性耐药性是治疗胃肠道间质瘤(GIST)的一大挑战。包括自噬在内的致癌通路的异常与伊马替尼耐药性有关。泛素-蛋白酶体系统对自噬相关蛋白 5(ATG5)的调控对自噬活性至关重要,但支持 ATG5 可逆去泛素化的分子机制尚未完全破解。在这里,我们发现 USP13 是一种重要的去泛素化酶,它能在一个依赖于 PAK1 丝氨酸/苏氨酸蛋白激酶的过程中稳定 ATG5,并能增强 GIST 细胞的自噬作用和促进 IM 抗性。USP13 在 GIST 细胞中被 IM 优先诱导,并与 ATG5 相互作用,从而通过去泛素化稳定 ATG5。PAK1 的激活促进了 ATG5 的磷酸化,从而增强了 ATG5 与 USP13 的相互作用。此外,N6-甲基腺苷甲基转移酶样 3(METTL3)介导了 USP13 mRNA 的稳定,这需要 m6A 阅读器 IGF2BP2。此外,USP13的抑制剂会导致ATG5衰减,在源自GIST细胞的小鼠异种移植模型中,该抑制剂与3-甲基腺嘌呤联合用药可提高IM的疗效。我们的研究结果突显了 USP13 是 GIST 细胞自噬和 IM 抗性的重要调节因子,并揭示了 USP13 是治疗 GIST 的潜在新靶点。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Cell Death and Differentiation
生物-生化与分子生物学
CiteScore
24.70
自引率
1.60%
发文量
181
审稿时长
3 months
期刊介绍:
Mission, vision and values of Cell Death & Differentiation:
To devote itself to scientific excellence in the field of cell biology, molecular biology, and biochemistry of cell death and disease.
To provide a unified forum for scientists and clinical researchers
It is committed to the rapid publication of high quality original papers relating to these subjects, together with topical, usually solicited, reviews, meeting reports, editorial correspondence and occasional commentaries on controversial and scientifically informative issues.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


