Wei Wang, Xiaohui Zhu, Xuecong Zhang, Chengyong Lei, Zhicheng Zeng, Xiaoliang Lan, Wenzhi Cui, Feifei Wang, Shaowan Xu, Juan Zhou, Xuehui Wu, Haijun Deng, Xia Li, Jianbing Fan, Yanqing Ding, Zhongxi Huang, Li Liang
下载PDF
{"title":"Recurrence risk assessment for stage III colorectal cancer based on five methylation biomarkers in plasma cell-free DNA","authors":"Wei Wang, Xiaohui Zhu, Xuecong Zhang, Chengyong Lei, Zhicheng Zeng, Xiaoliang Lan, Wenzhi Cui, Feifei Wang, Shaowan Xu, Juan Zhou, Xuehui Wu, Haijun Deng, Xia Li, Jianbing Fan, Yanqing Ding, Zhongxi Huang, Li Liang","doi":"10.1002/path.6047","DOIUrl":null,"url":null,"abstract":"<p>For stage III colorectal cancer (CRC) patients with a high risk of recurrence, intensified adjuvant chemotherapy can improve overall survival. We aimed to develop a circulating tumor DNA (ctDNA) methylation marker model for predicting the relapse risk of stage III CRC patients. Differentially methylated markers identified between 53 normal mucosa samples and 165 CRC tissue samples, as well as between plasma samples from 75 stage I/II (early-stage) CRC patients and 55 stage IV (late-stage) CRC patients, were analyzed using Student's <i>t</i>-tests. The overlapping methylation markers shared by plasma and tissue samples were used to establish a methylation marker model to evaluate the tumor burden in the peripheral blood of CRC patients using the random forest method. This model was verified in the validation cohort (<i>n</i> = 44) and then applied to predict recurrence risk in 50 stage III CRC patients and monitor the clinical disease course in serial samples from four CRC patients. We built a five-marker-based ctDNA methylation model that had high sensitivity (84.21%) and specificity (84%) in identifying late-stage CRC in a validation cohort containing 24 stage I/II CRC patients and 20 stage IV CRC patients. The model achieved high sensitivity (87.5%) and specificity (94.12%) in predicting tumor relapse in an independent cohort of 50 stage III CRC patients and could be an independent recurrence risk factor for stage III patients [Hazard ratio (HR), 60.4; 95% confidence interval (CI): 7.68–397; <i>p</i> = 9.73e−5]. Analysis of serial blood samples of CRC showed that the model could monitor disease relapse earlier than imaging examination and serum carcinoembryonic antigen (CEA) and so may provide an opportunity for the early adjustment of therapeutic strategies. Moreover, the model could potentially monitor the clinical course and treatment response dynamically. © 2022 The Authors. <i>The Journal of Pathology</i> published by John Wiley & Sons Ltd on behalf of The Pathological Society of Great Britain and Ireland.</p>","PeriodicalId":232,"journal":{"name":"The Journal of Pathology","volume":"259 4","pages":"376-387"},"PeriodicalIF":5.6000,"publicationDate":"2022-12-27","publicationTypes":"Journal Article","fieldsOfStudy":null,"isOpenAccess":false,"openAccessPdf":"https://onlinelibrary.wiley.com/doi/epdf/10.1002/path.6047","citationCount":"1","resultStr":null,"platform":"Semanticscholar","paperid":null,"PeriodicalName":"The Journal of Pathology","FirstCategoryId":"3","ListUrlMain":"https://onlinelibrary.wiley.com/doi/10.1002/path.6047","RegionNum":2,"RegionCategory":"医学","ArticlePicture":[],"TitleCN":null,"AbstractTextCN":null,"PMCID":null,"EPubDate":"","PubModel":"","JCR":"Q1","JCRName":"ONCOLOGY","Score":null,"Total":0}
引用次数: 1
引用
批量引用
Abstract
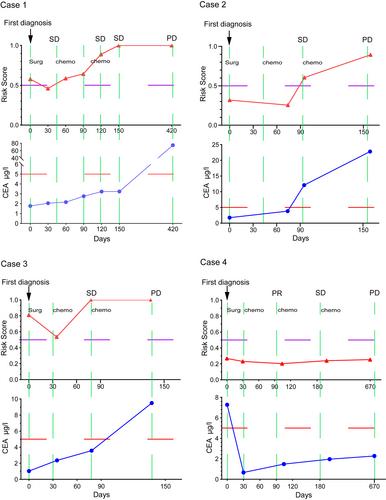
For stage III colorectal cancer (CRC) patients with a high risk of recurrence, intensified adjuvant chemotherapy can improve overall survival. We aimed to develop a circulating tumor DNA (ctDNA) methylation marker model for predicting the relapse risk of stage III CRC patients. Differentially methylated markers identified between 53 normal mucosa samples and 165 CRC tissue samples, as well as between plasma samples from 75 stage I/II (early-stage) CRC patients and 55 stage IV (late-stage) CRC patients, were analyzed using Student's t -tests. The overlapping methylation markers shared by plasma and tissue samples were used to establish a methylation marker model to evaluate the tumor burden in the peripheral blood of CRC patients using the random forest method. This model was verified in the validation cohort (n = 44) and then applied to predict recurrence risk in 50 stage III CRC patients and monitor the clinical disease course in serial samples from four CRC patients. We built a five-marker-based ctDNA methylation model that had high sensitivity (84.21%) and specificity (84%) in identifying late-stage CRC in a validation cohort containing 24 stage I/II CRC patients and 20 stage IV CRC patients. The model achieved high sensitivity (87.5%) and specificity (94.12%) in predicting tumor relapse in an independent cohort of 50 stage III CRC patients and could be an independent recurrence risk factor for stage III patients [Hazard ratio (HR), 60.4; 95% confidence interval (CI): 7.68–397; p = 9.73e−5]. Analysis of serial blood samples of CRC showed that the model could monitor disease relapse earlier than imaging examination and serum carcinoembryonic antigen (CEA) and so may provide an opportunity for the early adjustment of therapeutic strategies. Moreover, the model could potentially monitor the clinical course and treatment response dynamically. © 2022 The Authors. The Journal of Pathology published by John Wiley & Sons Ltd on behalf of The Pathological Society of Great Britain and Ireland.
基于血浆无细胞DNA中五种甲基化生物标志物的III期结直肠癌复发风险评估
对于具有高复发风险的III期癌症(CRC)患者,加强辅助化疗可以提高总体生存率。我们旨在开发一种循环肿瘤DNA(ctDNA)甲基化标记物模型,用于预测III期CRC患者的复发风险。使用Student t检验分析53个正常粘膜样本和165个CRC组织样本之间以及75个I/II期(早期)CRC患者和55个IV期(晚期)CRC患者的血浆样本之间鉴定的差异甲基化标记物。血浆和组织样本共享的重叠甲基化标记物用于建立甲基化标记模型,以使用随机森林法评估CRC患者外周血中的肿瘤负荷。该模型在验证队列中得到验证(n = 44),然后应用于预测50名III期CRC患者的复发风险,并在来自4名CRC患者的系列样本中监测临床病程。我们在一个包含24名I/II期CRC患者和20名IV期CRC患者的验证队列中建立了一个基于五个标记的ctDNA甲基化模型,该模型在识别晚期CRC方面具有高灵敏度(84.21%)和特异性(84%)。在50名III期CRC患者的独立队列中,该模型在预测肿瘤复发方面实现了高灵敏度(87.5%)和特异性(94.12%),并可能成为III期患者的独立复发风险因素[危险比(HR),60.4;95%置信区间(CI):7.68–397;p = 9.73e−5]。对CRC系列血样的分析表明,该模型可以比影像学检查和血清癌胚抗原(CEA)更早地监测疾病复发,从而为早期调整治疗策略提供了机会。此外,该模型可以动态监测临床过程和治疗反应。©2022作者。病理学杂志由John Wiley&;代表大不列颠及爱尔兰病理学会的Sons有限公司。
本文章由计算机程序翻译,如有差异,请以英文原文为准。

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


