Donor-Acceptor Copolymers with Rationally Regulated Side Chains Orientation for Polymer Solar Cells Processed by Non-Halogenated Solvent
引用次数: 2
Abstract
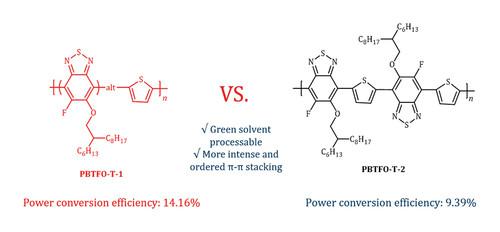
A donor-acceptor (D-A) conjugated polymer PBTFO-T-1 consisting of 2,1,3-benzothiadiazole (BT) as A unit and thiophene (T) as D unit was facilely obtained by a straightforward three-step reaction. The BT unit is attached with a fluorine atom and an alkoxy chain to simultaneously endow the polymer with a deep highest occupied molecular orbital (HOMO) energy level and desirable solubility. The alkoxyl chains orientation on BT unit has been regulated and the polymer PBTFO-T-2 with regio-regularly oriented side chains was also developed to investigate the impact of the alkoxyl chains orientation on their optoelectronic properties. The PBTFO-T-1: Y6-BO polymer solar cells (PSCs) were processed with non-halogenated solvent and achieve an optimized power conversion efficiency (PCE) of 14.16%, significantly higher than 9.39% of PBTFO-T-2: Y6-BO counterpart. It has been demonstrated that PBTFO-T-1: Y6-BO film exhibits higher and more balanced charge transportation and superior film morphology, resulting in higher exciton generation and dissociation, less recombination and eventually the higher short current density (Jsc) and fill factor (FF). This study provides a possible strategy to develop polymer donors with low cost for future commercial applications of PSCs and gives some insights on regulating optoelectronic properties of polymer donors via rationally modifying their side chains orientation.
非卤化溶剂处理聚合物太阳能电池中侧链取向合理调控的供体-受体共聚物
以2,1,3-苯并噻吩(T)为D单元,以A单元为2,1,3-苯并噻吩(BT)为A单元的给受体共轭聚合物PBTFO-T-1通过简单的三步反应得到。BT单元与氟原子和烷氧基链连接,以同时赋予聚合物具有深的最高已占据分子轨道(HOMO)能级和理想的溶解度。调控了烷氧基链在BT单元上的取向,并开发了具有区域规则取向侧链的聚合物PBTFO-T-2,研究了烷氧基链取向对其光电性能的影响。采用非卤化溶剂制备PBTFO-T-1: Y6-BO聚合物太阳能电池(PSCs),其优化功率转换效率(PCE)为14.16%,显著高于PBTFO-T-2: Y6-BO的9.39%。结果表明,PBTFO-T-1: Y6-BO薄膜表现出更高、更平衡的电荷输运和优越的薄膜形态,导致激子产生和解离更高,重组更少,最终获得更高的短电流密度(Jsc)和填充因子(FF)。该研究为未来PSCs的商业应用提供了低成本聚合物供体的可能策略,并为通过合理改变聚合物供体侧链取向来调节聚合物供体的光电性能提供了一些见解。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


