Preclinical to clinical translation for intervertebral disc repair: Effects of species-specific scale, metabolism, and matrix synthesis rates on cell-based regeneration
Abstract
Background
A significant hurdle for potential cell-based therapies is the subsequent survival and regenerative capacity of implanted cells. While many exciting developments have demonstrated promise preclinically, cell-based therapies for intervertebral disc (IVD) degeneration fail to translate equivalent clinical efficacy.
Aims
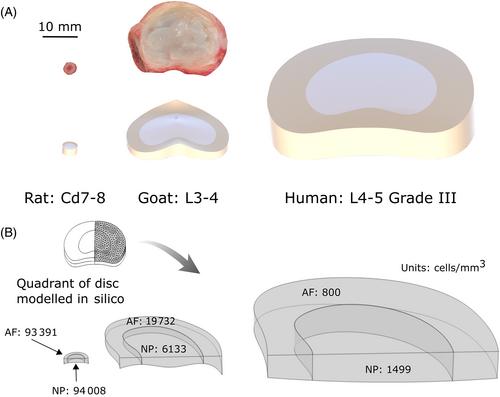
This work aims to ascertain the clinical relevance of both a small and large animal model by experimentally investigating and comparing these animal models to human from the perspective of anatomical scale and their cellular metabolic and regenerative potential.
Materials and Methods
First, this work experimentally investigated species-specific geometrical scale, native cell density, nutrient metabolism, and matrix synthesis rates for rat, goat, and human disc cells in a 3D microspheroid configuration. Second, these parameters were employed in silico to elucidate species-specific nutrient microenvironments and predict differences in temporal regeneration between animal models.
Results
This work presents in silico models which correlate favorably to preclinical literature in terms of the capabilities of animal regeneration and predict that compromised nutrition is not a significant challenge in small animal discs. On the contrary, it highlights a very fine clinical balance between an adequate cell dose for sufficient repair, through de novo matrix deposition, without exacerbating the human microenvironmental niche.
Discussion
Overall, this work aims to provide a path towards understanding the effect of cell injection number on the nutrient microenvironment and the “time to regeneration” between preclinical animal models and the large human IVD. While these findings help to explain failed translation of promising preclinical data and the limited results emerging from clinical trials at present, they also enable the research field and clinicians to manage expectations on cell-based regeneration.
Conclusion
Ultimately, this work provides a platform to inform the design of clinical trials, and as computing power and software capabilities increase in the future, it is conceivable that generation of patient-specific models could be used for patient assessment, as well as pre- and intraoperative planning.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


