Small changes in rhizosphere microbiome composition predict disease outcomes earlier than pathogen density variations
IF 10.8
1区 环境科学与生态学
Q1 ECOLOGY
引用次数: 30
Abstract
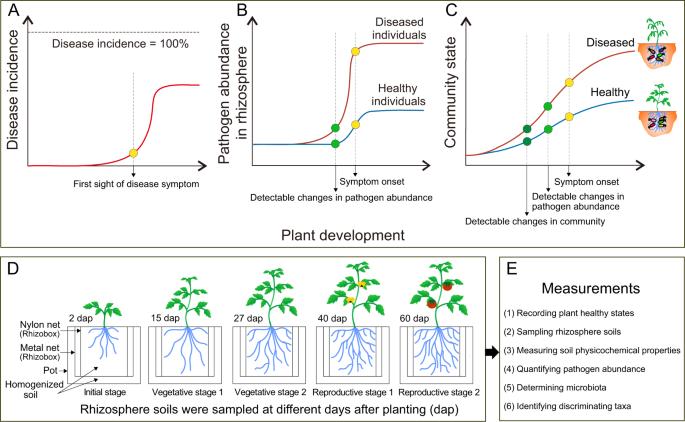
Even in homogeneous conditions, plants facing a soilborne pathogen tend to show a binary outcome with individuals either remaining fully healthy or developing severe to lethal disease symptoms. As the rhizosphere microbiome is a major determinant of plant health, we postulated that such a binary outcome may result from an early divergence in the rhizosphere microbiome assembly that may further cascade into varying disease suppression abilities. We tested this hypothesis by setting up a longitudinal study of tomato plants growing in a natural but homogenized soil infested with the soilborne bacterial pathogen Ralstonia solanacearum. Starting from an originally identical species pool, individual rhizosphere microbiome compositions rapidly diverged into multiple configurations during the plant vegetative growth. This variation in community composition was strongly associated with later disease development during the later fruiting state. Most interestingly, these patterns also significantly predicted disease outcomes 2 weeks before any difference in pathogen density became apparent between the healthy and diseased groups. In this system, a total of 135 bacterial OTUs were associated with persistent healthy plants. Five of these enriched OTUs (Lysinibacillus, Pseudarthrobacter, Bordetella, Bacillus, and Chryseobacterium) were isolated and shown to reduce disease severity by 30.4–100% when co-introduced with the pathogen. Overall, our results demonstrated that an initially homogenized soil can rapidly diverge into rhizosphere microbiomes varying in their ability to promote plant protection. This suggests that early life interventions may have significant effects on later microbiome states, and highlights an exciting opportunity for microbiome diagnostics and plant disease prevention.
根瘤菌群微生物组成的微小变化比病原体密度变化更早预测疾病结果
即使在同质条件下,植物在面对土传病原体时也往往会出现二元结果,要么完全健康,要么出现严重甚至致命的疾病症状。由于根圈微生物组是植物健康的主要决定因素,我们推测这种二元结果可能是根圈微生物组的早期分化造成的,这种分化可能进一步导致不同的疾病抑制能力。我们对生长在被土传细菌病原体 Ralstonia solanacearum 侵染的天然均质土壤中的番茄植株进行了纵向研究,从而验证了这一假设。从最初完全相同的物种库开始,在植物无性生长过程中,各个根瘤微生物群组成迅速分化成多种配置。群落组成的这种变化与后期结果期的病害发展密切相关。最有趣的是,在病原体密度在健康组和患病组之间出现明显差异之前两周,这些模式还能显著预测病害结果。在该系统中,共有 135 个细菌 OTU 与持续健康的植株相关。在这些富集的 OTU 中,有 5 个(赖氨巴氏杆菌、假节杆菌、博德特氏菌、芽孢杆菌和绿脓杆菌)被分离出来,并被证明在与病原体共同引入时可将病害严重程度降低 30.4%-100%。总之,我们的研究结果表明,最初均质的土壤会迅速分化成根瘤微生物群,这些微生物群促进植物保护的能力各不相同。这表明,生命早期的干预措施可能会对后期的微生物组状态产生重大影响,并为微生物组诊断和植物病害预防带来了令人兴奋的机遇。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

ISME Journal
环境科学-生态学
CiteScore
22.10
自引率
2.70%
发文量
171
审稿时长
2.6 months
期刊介绍:
The ISME Journal covers the diverse and integrated areas of microbial ecology. We encourage contributions that represent major advances for the study of microbial ecosystems, communities, and interactions of microorganisms in the environment. Articles in The ISME Journal describe pioneering discoveries of wide appeal that enhance our understanding of functional and mechanistic relationships among microorganisms, their communities, and their habitats.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


