Dual nickel/photoredox catalyzed carboxylation of C(sp2)-halides with formate†
Organic chemistry frontiers : an international journal of organic chemistry
Pub Date : 2023-01-01
DOI:10.1039/d2qo01361d
引用次数: 3
Abstract
Herein, we report an efficient and practical protocol for dehalocarboxylation of C(sp2)-halides with formate by the engagement of a CO2 radical anion in a nickel-mediated bond-forming process. A wide variety of aryl iodides, aryl bromides, and alkenyl bromides bearing a diverse set of functional groups underwent the reaction smoothly through visible-light photoredox nickel dual catalysis. The synthesis of several 13C-labeled drug intermediates and the gram-scale synthesis of commercial drugs highlight the synthetic value of the approach in drug discovery settings.
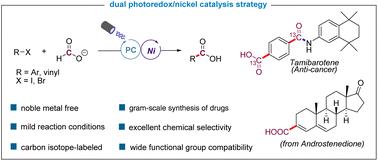
双镍/光氧化还原催化C(sp2)-卤化物与甲酸†的羧化反应
在此,我们报道了一种高效实用的方案,通过在镍介导的键形成过程中与CO2自由基阴离子结合,用甲酸盐对C(sp2)-卤化物进行脱卤羧化。多种具有不同官能团的芳基碘化物、芳基溴化物和链烯基溴化物通过可见光-光还原镍双催化顺利进行反应。几种13C标记的药物中间体的合成和商业药物的克级合成突出了该方法在药物发现环境中的合成价值。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
7.80
自引率
0.00%
发文量
0

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


