Synthesis of cyclopenta[b]benzofurans via biomimetic oxidative phenol–enamine [3 + 2] cycloaddition†
Organic chemistry frontiers : an international journal of organic chemistry
Pub Date : 2023-01-01
DOI:10.1039/d2qo01916g
引用次数: 0
Abstract
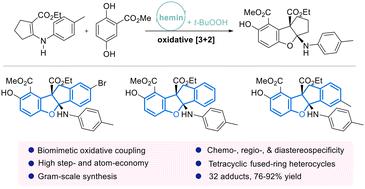
A hemin/t-BuOOH-catalyzed oxidative phenol–enamine formal [3 + 2] cycloaddition has been developed, enabling the environmentally benign synthesis of valuable cyclopenta[b]benzofuran scaffolds under sustainable reaction conditions with excellent atom- and step-economy. Mechanistic studies point to the involvement of a two-electron oxidation pathway. Based on the preliminary biological investigations, compound 3ay displayed moderate anti-cancer activities (IC50 = 27.73 μM) against MCF7 cells (breast cancer cells).
仿生氧化苯酚- enamine[3 + 2]环加成法合成环五[b]苯并呋喃
已经开发了一种血红素/叔丁醇催化的氧化苯酚-烯胺形式[3+2]环加成,使其能够在可持续的反应条件下以优异的原子经济性和步进经济性对环境无害地合成有价值的环戊基[b]苯并呋喃支架。机制学研究表明,这与双电子氧化途径有关。根据初步的生物学研究,化合物3ay对MCF7细胞(癌症细胞)表现出中等的抗癌活性(IC50=27.73μM)。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
7.80
自引率
0.00%
发文量
0

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


