A general protocol for stereoselective construction of enaminyl sulfonyl fluorides†
Organic chemistry frontiers : an international journal of organic chemistry
Pub Date : 2023-01-01
DOI:10.1039/d2qo01810a
引用次数: 1
Abstract
A simple, efficient, and mild reaction of 2-chloroprop-2-ene-1-sulfonyl fluoride (CESF) with amines was developed, affording a variety of highly functionalized enaminyl sulfonyl fluorides in good to excellent yields (56–96%). This transformation features a broad substrate scope, high atom economy, operational simplicity and up to 100% stereoselectivity, which can provide great application value in medicinal chemistry, chemical biology, and drug discovery.
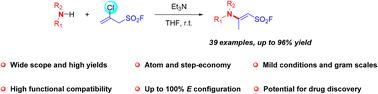
烯甲酰基磺酰基氟化物立体选择性构建的通用方案†
开发了一种简单、高效、温和的2-氯丙基-2-烯-1-磺酰基氟化物(CESF)与胺的反应,以良好至优异的产率(56–96%)提供了各种高度官能化的烯酰基磺酰基氟化物。该转化具有底物范围广、原子经济性高、操作简单、立体选择性高达100%的特点,在药物化学、化学生物学和药物发现方面具有巨大的应用价值。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
7.80
自引率
0.00%
发文量
0

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


