Palladium-catalyzed intramolecular C–H amination using aluminum nitrate as the oxidant†
Organic chemistry frontiers : an international journal of organic chemistry
Pub Date : 2023-01-01
DOI:10.1039/d2qo01562e
引用次数: 0
Abstract
A palladium catalyzed intramolecular C(sp2)–H amination using a readily available aluminum nitrate (Al(NO3)3·9H2O) as the oxidant is reported. The C–H amination is promoted by in situ nitration of the quinoline directing group (DG). The oxindole products can be efficiently converted into novel polycyclic compounds (up to 7 fused rings) via a sequential reduction/cyclization involving the DG. Mechanistic studies indicate that nitration of the quinoline DG favors the following activation of γ-aryl C(sp2)–H bonds over β-methyl C(sp3)–H bonds.
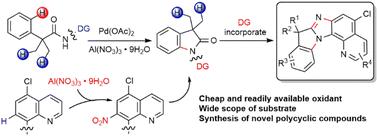
钯催化的分子内C–H胺化,使用硝酸铝作为氧化剂†
报道了以易得的硝酸铝(Al(NO3)3·9H2O)为氧化剂,钯催化的分子内C(sp2)–H胺化反应。喹啉导向基团(DG)的原位硝化促进了C–H胺化。羟吲哚产物可以通过涉及DG的顺序还原/环化有效地转化为新的多环化合物(最多7个稠环)。机理研究表明,喹啉DG的硝化有利于γ-芳基C(sp2)–H键的后续活化,而不是β-甲基C(sp3)–H。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
7.80
自引率
0.00%
发文量
0

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


