A reductive Sandmeyer-type reaction for the synthesis of sulfoxides from anilines under photocatalysis†
Organic chemistry frontiers : an international journal of organic chemistry
Pub Date : 2023-01-01
DOI:10.1039/d3qo00297g
引用次数: 0
Abstract
A reductive Sandmeyer-type sulfinylation reaction of aryldiazonium salts with sodium sulfinates is reported. The reaction takes place under photocatalysis, generating a range of valuable sulfoxides via a radical substitution pathway. Aromatic amines can also be converted to the corresponding sulfoxides via in situ diazotization in a one-pot, two-step process. The late-stage sulfinylation of drug-based amines further demonstrated the practicality of this method.
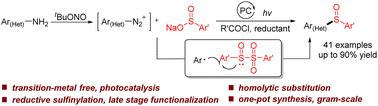
在光催化下由苯胺合成亚砜的还原Sandmeyer型反应†
报道了芳基二氮鎓盐与亚磺酸钠的还原Sandmeyer型亚磺酰基化反应。反应在光催化下进行,通过自由基取代途径产生一系列有价值的亚砜。芳香胺也可以通过一锅两步法原位重氮化转化为相应的亚砜。药物胺的后期亚磺酰基化进一步证明了该方法的实用性。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
7.80
自引率
0.00%
发文量
0

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


