A novel feedback loop: CELF1/circ-CELF1/BRPF3/KAT7 in cardiac fibrosis
IF 14.6
1区 医学
Q1 PHARMACOLOGY & PHARMACY
引用次数: 0
Abstract
Cardiac fibrosis is characterized by an elevated amount of extracellular matrix (ECM) within the heart. However, the persistence of cardiac fibrosis ultimately diminishes contractility and precipitates cardiac dysfunction. Circular RNAs (circRNAs) are emerging as important regulators of cardiac fibrosis. Here, we elucidate the functional role of a specific circular RNA CELF1 in cardiac fibrosis and delineate a novel feedback loop mechanism. Functionally, circ-CELF1 was involved in enhancing fibrosis-related markers' expression and promoting the proliferation of cardiac fibroblasts (CFs), thereby exacerbating cardiac fibrosis. Mechanistically, circ-CELF1 reduced the ubiquitination-degradation rate of BRPF3, leading to an elevation of BRPF3 protein levels. Additionally, BRPF3 acted as a modular scaffold for the recruitment of histone acetyltransferase KAT7 to facilitate the induction of H3K14 acetylation within the promoters of the Celf1 gene. Thus, the transcription of Celf1 was dramatically activated, thereby inhibiting the subsequent response of their downstream target gene Smad7 expression to promote cardiac fibrosis. Moreover, Celf1 further promoted Celf1 pre-mRNA transcription and back-splicing, thereby establishing a feedback loop for circ-CELF1 production. Consequently, a novel feedback loop involving CELF1/circ-CELF1/BRPF3/KAT7 was established, suggesting that circ-CELF1 may serve as a potential novel therapeutic target for cardiac fibrosis.
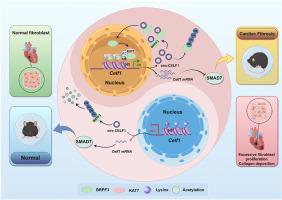
一个新的反馈回路:CELF1/circ-CELF1/BRPF3/KAT7在心脏纤维化中的作用
心脏纤维化的特征是心脏内细胞外基质(ECM)含量升高。然而,心脏纤维化的持续存在最终会降低心脏的收缩力并导致心功能障碍。环状rna (circRNAs)正在成为心脏纤维化的重要调节因子。在这里,我们阐明了一种特殊的环状RNA CELF1在心脏纤维化中的功能作用,并描绘了一种新的反馈回路机制。在功能上,circ-CELF1参与增强纤维化相关标志物的表达,促进心脏成纤维细胞(CFs)的增殖,从而加剧心脏纤维化。从机制上讲,circ-CELF1降低了BRPF3的泛素化降解率,导致BRPF3蛋白水平升高。此外,BRPF3作为组蛋白乙酰转移酶KAT7募集的模块化支架,促进在Celf1基因启动子内诱导H3K14乙酰化。因此,Celf1的转录被显著激活,从而抑制其下游靶基因Smad7表达的后续反应,促进心脏纤维化。此外,Celf1进一步促进了Celf1 pre-mRNA转录和反剪接,从而建立了circ-CELF1产生的反馈回路。因此,建立了一个涉及CELF1/circ-CELF1/BRPF3/KAT7的新型反馈回路,表明circ-CELF1可能作为心脏纤维化的潜在新型治疗靶点。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Acta Pharmaceutica Sinica. B
Pharmacology, Toxicology and Pharmaceutics-General Pharmacology, Toxicology and Pharmaceutics
CiteScore
22.40
自引率
5.50%
发文量
1051
审稿时长
19 weeks
期刊介绍:
The Journal of the Institute of Materia Medica, Chinese Academy of Medical Sciences, and the Chinese Pharmaceutical Association oversees the peer review process for Acta Pharmaceutica Sinica. B (APSB).
Published monthly in English, APSB is dedicated to disseminating significant original research articles, rapid communications, and high-quality reviews that highlight recent advances across various pharmaceutical sciences domains. These encompass pharmacology, pharmaceutics, medicinal chemistry, natural products, pharmacognosy, pharmaceutical analysis, and pharmacokinetics.
A part of the Acta Pharmaceutica Sinica series, established in 1953 and indexed in prominent databases like Chemical Abstracts, Index Medicus, SciFinder Scholar, Biological Abstracts, International Pharmaceutical Abstracts, Cambridge Scientific Abstracts, and Current Bibliography on Science and Technology, APSB is sponsored by the Institute of Materia Medica, Chinese Academy of Medical Sciences, and the Chinese Pharmaceutical Association. Its production and hosting are facilitated by Elsevier B.V. This collaborative effort ensures APSB's commitment to delivering valuable contributions to the pharmaceutical sciences community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


