A reflection on ketoABNO: the crossing point between organic synthesis and protein modification
IF 7.4
1区 化学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
In 2012, we reported that the N-oxyl radical ketoABNO functions as an effective catalyst for the mild aerobic oxidation of amines to imines (T. Sonobe, K. Oisaki and M. Kanai, Chem. Sci., 2012, 3, 3249, https://doi.org/10.1039/C2SC20699D). Its catalytic versatility arises from a unique combination of steric compactness, high oxidation potential, and the ability to reversibly interconvert among three oxidation states—hydroxyamine, N-oxyl, and oxoammonium. Beyond amine oxidation, ketoABNO has also been applied to the oxidation of alcohols and aldehydes. More recently, its utility has extended beyond small-molecule transformations to include applications in protein modifications, such as serine-selective oxidative cleavage of proteins (in conjunction with a water-soluble copper-complex catalyst) and tryptophan-selective bioconjugation. In this Commentary, we highlight the development of ketoABNO as an oxidation catalyst and its emerging applications in biocompatible protein chemistry.
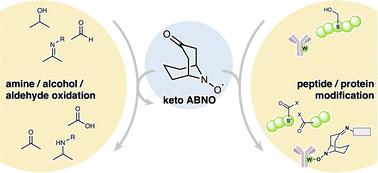
酮abno:有机合成与蛋白质修饰的交叉点
在2012年,我们报道了n -氧自由基ketoABNO作为一种有效的催化剂,可以将胺轻度有氧氧化为亚胺(T. Sonobe, K. Oisaki和M. Kanai, Chem.)。科学。[j], 2012, 3, 3249, https://doi.org/10.1039/C2SC20699D)。它的催化多功能性源于其独特的空间紧致性、高氧化电位以及在三种氧化态(羟胺、n -氧和氧铵)之间可逆相互转化的能力。除了胺氧化,酮abno也被应用于醇和醛的氧化。最近,它的用途已经从小分子转化扩展到蛋白质修饰,例如蛋白质的丝氨酸选择性氧化切割(与水溶性铜络合物催化剂结合)和色氨酸选择性生物偶联。在这篇评论中,我们重点介绍了酮abno作为氧化催化剂的发展及其在生物相容性蛋白质化学中的新应用。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Chemical Science
CHEMISTRY, MULTIDISCIPLINARY-
CiteScore
14.40
自引率
4.80%
发文量
1352
审稿时长
2.1 months
期刊介绍:
Chemical Science is a journal that encompasses various disciplines within the chemical sciences. Its scope includes publishing ground-breaking research with significant implications for its respective field, as well as appealing to a wider audience in related areas. To be considered for publication, articles must showcase innovative and original advances in their field of study and be presented in a manner that is understandable to scientists from diverse backgrounds. However, the journal generally does not publish highly specialized research.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


