Nickel-catalyzed [4 + 1] sila-cycloaddition: a divergent synthesis of silacarbocycles from trichlorosilanes
IF 4.7
1区 化学
Q1 CHEMISTRY, ORGANIC
引用次数: 0
Abstract
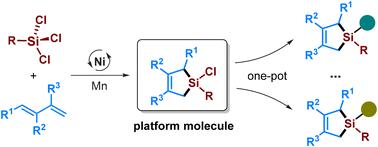
Cyclic chlorosilane transformations present a valuable opportunity for the divergent synthesis of silacarbocycles, but their low availability has hindered this potential. This study presents a direct approach to producing structurally diverse five-membered cyclic chlorosilanes, utilizing trichlorosilanes as a novel Si-1 synthon for catalytic sila-cycloaddition reactions. The method employs a nickel-catalyzed [4 + 1] cycloaddition reaction between 1,3-dienes and trichlorosilanes, facilitated by upgraded phosphine–nitrogen ligands. This approach accommodates a broad range of 1,3-dienes and trichlorosilanes, yielding cyclic chlorosilanes that act as versatile platform molecules. These intermediates can be readily transformed into advanced silicon reagents and connected with various carbon and heteroatom functionalities through a one-pot reaction.
镍催化的[4 + 1]硅环加成:三氯硅烷发散合成硅碳环
环氯硅烷转化为硅碳环的发散合成提供了宝贵的机会,但它们的低可用性阻碍了这一潜力。本研究提出了一种直接制备结构多样的五元环氯硅烷的方法,利用三氯硅烷作为催化硅-环加成反应的新型Si-1合成物。该方法采用镍催化的1,3-二烯与三氯硅烷之间的[4 + 1]环加成反应,通过升级的膦-氮配体促进。这种方法适用于广泛的1,3-二烯和三氯硅烷,产生作为多功能平台分子的环氯硅烷。这些中间体可以很容易地转化为先进的硅试剂,并通过一锅反应与各种碳和杂原子官能团连接。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Organic Chemistry Frontiers
CHEMISTRY, ORGANIC-
CiteScore
7.90
自引率
11.10%
发文量
686
审稿时长
1 months
期刊介绍:
Organic Chemistry Frontiers is an esteemed journal that publishes high-quality research across the field of organic chemistry. It places a significant emphasis on studies that contribute substantially to the field by introducing new or significantly improved protocols and methodologies. The journal covers a wide array of topics which include, but are not limited to, organic synthesis, the development of synthetic methodologies, catalysis, natural products, functional organic materials, supramolecular and macromolecular chemistry, as well as physical and computational organic chemistry.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


