Targeting tumor monocyte-intrinsic PD-L1 by rewiring STING signaling and enhancing STING agonist therapy
IF 48.8
1区 医学
Q1 CELL BIOLOGY
引用次数: 0
Abstract
STING is an important DNA sensing machinery in initiating immune response, yet therapies targeting STING have shown poor outcomes in clinical trials. Here, we reveal that STING signaling induces PD-L1hi tumor monocytes (Tu.Mons) that dominate the resistance against STING agonist therapy. Cell-intrinsic PD-L1, induced by the STING-IRF3-IFN-I axis, is identified as the driving factor for protumoral PD-L1hi Tu.Mons. Notably, TLR2-activated Tu.Mons resist STING-induced upregulation of cell-intrinsic PD-L1 and the associated protumoral functions. Mechanistically, TLR2 stimulation remodels STING signaling by facilitating STING and TRAF6 interaction, which suppresses the IRF3-IFN-I response and enhances NF-κB activation. Moreover, we demonstrate that combining STING agonists with TLR2 agonist pretreatment significantly improves antitumor efficacy in murine syngeneic and humanized models. Our findings uncover a protumoral aspect of STING activation mediated by cell-intrinsic PD-L1 and propose a promising strategy to boost antitumor immunity by fine-tuning STING signaling outputs.
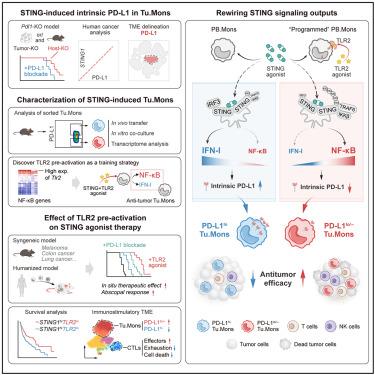
通过重新连接STING信号和增强STING激动剂治疗靶向肿瘤单核细胞内生性PD-L1
STING是启动免疫应答的重要DNA传感机制,但针对STING的治疗在临床试验中表现出较差的效果。在这里,我们揭示了STING信号诱导PD-L1hi肿瘤单核细胞(Tu.Mons)主导对STING激动剂治疗的抗性。由STING-IRF3-IFN-I轴诱导的细胞内禀PD-L1被认为是PD-L1肿瘤发生的驱动因子。值得注意的是,tlr2激活的tumon可抵抗sting诱导的细胞内禀PD-L1上调和相关的肿瘤功能。从机制上讲,TLR2刺激通过促进STING和TRAF6相互作用来重塑STING信号,从而抑制IRF3-IFN-I反应并增强NF-κB激活。此外,我们证明STING激动剂与TLR2激动剂预处理联合使用可显著提高小鼠同源和人源化模型的抗肿瘤效果。我们的研究结果揭示了细胞内禀PD-L1介导的STING激活的原发肿瘤方面,并提出了一种通过微调STING信号输出来增强抗肿瘤免疫的有希望的策略。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Cancer Cell
医学-肿瘤学
CiteScore
55.20
自引率
1.20%
发文量
179
审稿时长
4-8 weeks
期刊介绍:
Cancer Cell is a journal that focuses on promoting major advances in cancer research and oncology. The primary criteria for considering manuscripts are as follows:
Major advances: Manuscripts should provide significant advancements in answering important questions related to naturally occurring cancers.
Translational research: The journal welcomes translational research, which involves the application of basic scientific findings to human health and clinical practice.
Clinical investigations: Cancer Cell is interested in publishing clinical investigations that contribute to establishing new paradigms in the treatment, diagnosis, or prevention of cancers.
Insights into cancer biology: The journal values clinical investigations that provide important insights into cancer biology beyond what has been revealed by preclinical studies.
Mechanism-based proof-of-principle studies: Cancer Cell encourages the publication of mechanism-based proof-of-principle clinical studies, which demonstrate the feasibility of a specific therapeutic approach or diagnostic test.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


