Lymph node macrophages drive immune tolerance and resistance to cancer therapy by induction of the immune-regulatory cytokine IL-33
IF 48.8
1区 医学
Q1 CELL BIOLOGY
引用次数: 0
Abstract
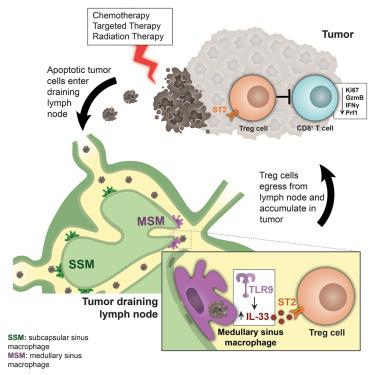
Apoptotic cells are immunosuppressive, creating a barrier in cancer treatment. Thus, we investigated immune responses to dying tumor cells after therapy in the tumor draining lymph node (TDLN). A key population responsible for clearing tumor material in the TDLN was medullary sinus macrophages (MSMs). Tumor debris phagocytosis by MSMs induces the cytokine IL-33, and blocking the IL-33 receptor (ST2) or deletion of Il33 in MSMs enhances therapy responses. Mechanistically, IL-33 activates T regulatory cells in TDLNs that migrate to the tumor to suppress CD8+ T cells. Therapeutically combining ST2 blockade, targeted kinase inhibitors, and anti-PD-1 immunotherapy increases CD8+ T cell activity promoting tumor regression. Importantly, we observe similar activity in human macrophages, and IL-33 expression in sentinel lymph nodes correlates with disease stage and survival in melanoma. Thus, our data identifies an IL-33-dependent immune response to therapy that attenuates therapy-induced anti-tumor immunity.
求助全文
约1分钟内获得全文
求助全文
来源期刊

Cancer Cell
医学-肿瘤学
CiteScore
55.20
自引率
1.20%
发文量
179
审稿时长
4-8 weeks
期刊介绍:
Cancer Cell is a journal that focuses on promoting major advances in cancer research and oncology. The primary criteria for considering manuscripts are as follows:
Major advances: Manuscripts should provide significant advancements in answering important questions related to naturally occurring cancers.
Translational research: The journal welcomes translational research, which involves the application of basic scientific findings to human health and clinical practice.
Clinical investigations: Cancer Cell is interested in publishing clinical investigations that contribute to establishing new paradigms in the treatment, diagnosis, or prevention of cancers.
Insights into cancer biology: The journal values clinical investigations that provide important insights into cancer biology beyond what has been revealed by preclinical studies.
Mechanism-based proof-of-principle studies: Cancer Cell encourages the publication of mechanism-based proof-of-principle clinical studies, which demonstrate the feasibility of a specific therapeutic approach or diagnostic test.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


