Engineering Affibody Binders to Death Receptor 5 and Tumor Necrosis Factor Receptor 1 With Improved Stability
IF 3.5
2区 生物学
Q2 BIOTECHNOLOGY & APPLIED MICROBIOLOGY
引用次数: 0
Abstract
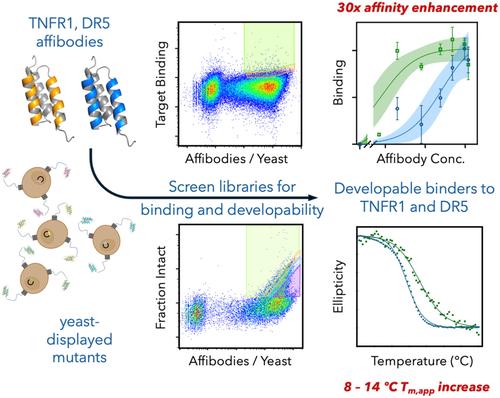
Protein developability is an important, yet often overlooked, aspect of protein discovery campaigns that is a key driver of utility. Recent advances have improved developability screening capacity, making it an increasingly viable option in early-stage discovery. Here, we engineered one component of developability, stability, of two affibody proteins—one that targets death receptor 5 and another that targets tumor necrosis factor receptor 1—previously evolved to bind receptor and non-competitively inhibit signaling via conformational modulation. Starting from an error-prone PCR library of each affibody, variants were screened via yeast surface display binder selections, including depletion of non-specific binders, followed by developability assessment using the on-yeast protease and yeast display level assays. Multiplex deep sequencing identified variants for further evaluation. Purified variants exhibited elevated stability—8°C to 14°C increase in Tm,app—with maintained 1–2 nM affinity for the TNFR1 affibody and 30-fold improvement in the DR5 affibody affinity to 0.8 nM.
求助全文
约1分钟内获得全文
求助全文
来源期刊

Biotechnology and Bioengineering
工程技术-生物工程与应用微生物
CiteScore
7.90
自引率
5.30%
发文量
280
审稿时长
2.1 months
期刊介绍:
Biotechnology & Bioengineering publishes Perspectives, Articles, Reviews, Mini-Reviews, and Communications to the Editor that embrace all aspects of biotechnology. These include:
-Enzyme systems and their applications, including enzyme reactors, purification, and applied aspects of protein engineering
-Animal-cell biotechnology, including media development
-Applied aspects of cellular physiology, metabolism, and energetics
-Biocatalysis and applied enzymology, including enzyme reactors, protein engineering, and nanobiotechnology
-Biothermodynamics
-Biofuels, including biomass and renewable resource engineering
-Biomaterials, including delivery systems and materials for tissue engineering
-Bioprocess engineering, including kinetics and modeling of biological systems, transport phenomena in bioreactors, bioreactor design, monitoring, and control
-Biosensors and instrumentation
-Computational and systems biology, including bioinformatics and genomic/proteomic studies
-Environmental biotechnology, including biofilms, algal systems, and bioremediation
-Metabolic and cellular engineering
-Plant-cell biotechnology
-Spectroscopic and other analytical techniques for biotechnological applications
-Synthetic biology
-Tissue engineering, stem-cell bioengineering, regenerative medicine, gene therapy and delivery systems
The editors will consider papers for publication based on novelty, their immediate or future impact on biotechnological processes, and their contribution to the advancement of biochemical engineering science. Submission of papers dealing with routine aspects of bioprocessing, description of established equipment, and routine applications of established methodologies (e.g., control strategies, modeling, experimental methods) is discouraged. Theoretical papers will be judged based on the novelty of the approach and their potential impact, or on their novel capability to predict and elucidate experimental observations.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


