Dual-functional RhNi nanocubes for coupling electrocatalytic water reduction and alcohol oxidation for energy-saving hydrogen production
IF 6.1
1区 化学
Q1 CHEMISTRY, INORGANIC & NUCLEAR
引用次数: 0
Abstract
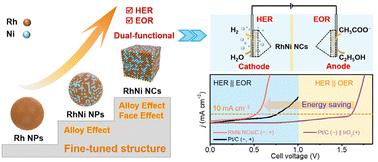
Electrochemical water splitting holds promise for sustainable hydrogen (H2) production, yet it faces the challenge of high energy demand due to significant overpotentials and sluggish kinetics for the anodic oxygen evolution reaction (OER). An efficient alternative is to replace the energy-intensive OER with the more thermodynamically advantageous alcohol oxidation reaction (AOR). Accordingly, catalysts that enhance both the hydrogen evolution reaction (HER) and the AOR may pave the way for sustainable and efficient hydrogen production. Herein, we demonstrate that the composition and morphology engineering of Rh–Ni alloy nanocrystals can significantly boost their dual functionality for the HER and the ethanol oxidation reaction (EOR). Theoretical and experimental analyses reveal the enhanced H2O dissociation, alcohol oxidation capacity, and CO tolerance of RhNi NCs with predominant {100} facets. As a result, a two-electrode electrolysis cell using RhNi NCs as dual-functional catalysts for the HER||EOR can achieve a current density of 10 mA cm−2 and 50 mA cm−2 at 0.53 V and 0.68 V, respectively, outperforming the Pt/C and IrO2 benchmark catalysts. Remarkably, the RhNi NCs can also catalyze the oxidation of a range of alcohols for energy-saving H2 production at low cell voltages. Our strategy represents a rational catalyst design achieved through controlled solution synthesis, holding potential for broader applicability in future electrocatalysis and clean energy conversion.
求助全文
约1分钟内获得全文
求助全文
来源期刊

Inorganic Chemistry Frontiers
CHEMISTRY, INORGANIC & NUCLEAR-
CiteScore
10.40
自引率
7.10%
发文量
587
审稿时长
1.2 months
期刊介绍:
The international, high quality journal for interdisciplinary research between inorganic chemistry and related subjects
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


