Monodentate σ-Accepting Boron-Based Ligands Bearing Square-Planar Ni(0) Centers
IF 14.4
1区 化学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
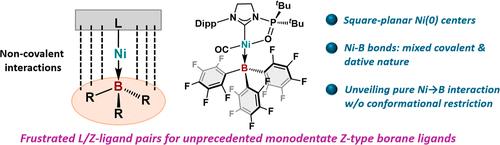
Transition metals are known to work as electron donors toward electron-accepting heavier-group-13 elements (Al, Ga, and In), called Z-type ligands. However, complexes with boron-based Z-type ligands are stable only in the presence of additional coordination units (the so-called “supported-ligand” strategy). Here, we report the synthesis and characterization of square-planar Ni(0) complexes that bear tris(perfluoroaryl)boranes as monodentate Z-type ligands, even though such coordination geometry has been traditionally associated with Ni(II) species based on the well-established ligand-field theory. A combined theoretical and experimental approach revealed a mixed covalent/dative character for the Ni–B bonds. This strategy uses frustrated L/Z-ligand pairs that combine sterically encumbered electron-donating (L-type) and electron-accepting ligands to form noncovalent interactions over L–M–Z units to achieve unprecedented low-valent transition metal species with monodentate Z-type ligands.
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
24.40
自引率
6.00%
发文量
2398
审稿时长
1.6 months
期刊介绍:
The flagship journal of the American Chemical Society, known as the Journal of the American Chemical Society (JACS), has been a prestigious publication since its establishment in 1879. It holds a preeminent position in the field of chemistry and related interdisciplinary sciences. JACS is committed to disseminating cutting-edge research papers, covering a wide range of topics, and encompasses approximately 19,000 pages of Articles, Communications, and Perspectives annually. With a weekly publication frequency, JACS plays a vital role in advancing the field of chemistry by providing essential research.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


