First-principles thermodynamic modeling of hydrogen dissolution in metals
IF 8.3
1区 材料科学
Q1 MATERIALS SCIENCE, MULTIDISCIPLINARY
引用次数: 0
Abstract
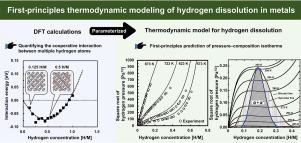
To efficiently explore and design alloys with excellent hydrogen permeability, it is critical to clarify the bottleneck steps in the hydrogen permeation process. Hydrogen dissolution is one of the most important elementary processes in hydrogen permeation, regarding not only hydrogen permeability but also the suppression of hydrogen embrittlement. In this paper, we propose an effective and robust thermodynamic model for hydrogen dissolution in metals that can precisely describe the pressure–composition isotherm (i.e., the PCT curve) over a wide range of hydrogen concentrations. With this model, the PCT curves for the V and V–Fe systems were described nonempirically in a manner consistent with their experimental counterparts using only parameters obtained via density functional theory (DFT) calculations. The essential factors that determine the hydrogen concentration dependence of hydrogen dissolution in metals are the cooperative interactions between multiple hydrogen atoms in the metal, including the site-blocking effect. By quantifying these factors using DFT calculations and introducing them into the model, it was confirmed that the hydrogen dissolution behavior deviated from Sieverts’ law, as observed for many metals and alloys. Furthermore, we found that the addition of Fe to V increased the interaction energy between the hydrogen atoms, reducing the solubility of hydrogen in V. Our DFT-informed model advances the understanding of the mechanism of hydrogen dissolution in metals and offers new insights into controlling hydrogen solubility at target temperatures to suppress hydrogen embrittlement in hydrogen-permeable metals. The findings of this study provide theoretical guidance for the efficient design of hydrogen-permeable and hydrogen-storage materials.

求助全文
约1分钟内获得全文
求助全文
来源期刊

Acta Materialia
工程技术-材料科学:综合
CiteScore
16.10
自引率
8.50%
发文量
801
审稿时长
53 days
期刊介绍:
Acta Materialia serves as a platform for publishing full-length, original papers and commissioned overviews that contribute to a profound understanding of the correlation between the processing, structure, and properties of inorganic materials. The journal seeks papers with high impact potential or those that significantly propel the field forward. The scope includes the atomic and molecular arrangements, chemical and electronic structures, and microstructure of materials, focusing on their mechanical or functional behavior across all length scales, including nanostructures.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


