Haploinsufficiency of miR-143 and miR-145 reveal targetable dependencies in resistant del(5q) myelodysplastic neoplasm
IF 12.8
1区 医学
Q1 HEMATOLOGY
引用次数: 0
Abstract
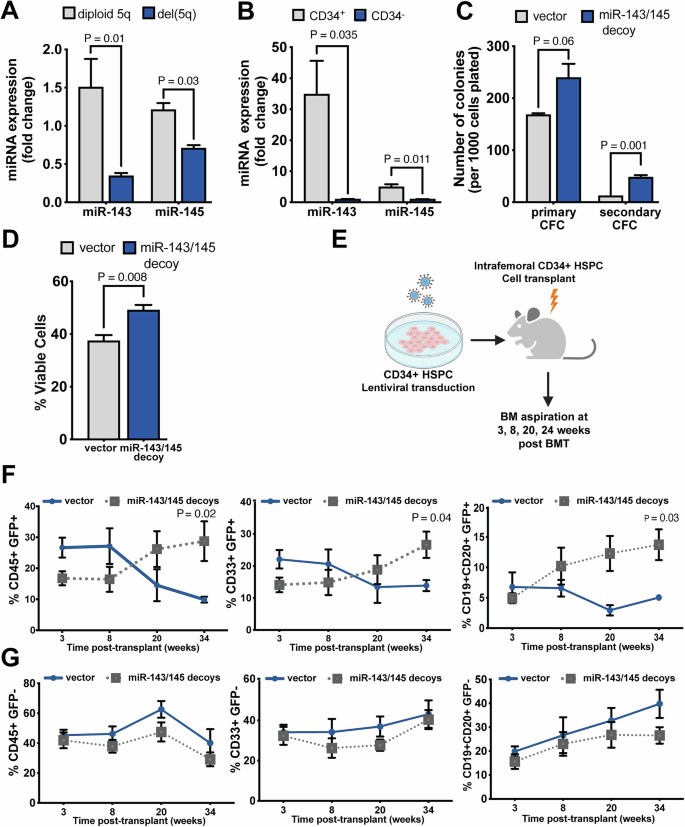
Myelodysplastic neoplasms (MDS) are stem cell disorders characterized by ineffective hematopoiesis and risk of transformation to acute myeloid leukemia (AML). Chromosomal alterations are frequent in MDS, with interstitial deletion of chromosome 5q (del(5q)) being the most common. Lenalidomide is the current first-line treatment for del(5q) MDS and its efficacy relies on degradation of CK1α which is encoded by the CSNK1A1 gene located in the commonly deleted region (CDR) of chromosome 5q. However, lenalidomide-resistance is common, often secondary to loss-of-function mutations in TP53 or RUNX1. The CDR in del(5q) harbors several genes, including noncoding miRNAs, the loss of which contribute to disease phenotypes. miR-143 and miR-145 are located within the del(5q) CDR, but precise understanding of their role in human hematopoiesis and in the pathogenesis of del(5q) MDS is lacking. Here we provide evidence that deficiency of miR-143 and miR-145 plays a role in clonal expansion of del(5q) MDS. We show that insulin-like growth factor 1 receptor (IGF-1R) is a direct target of both miR-143 and miR-145. Our data demonstrate that IGF-1R inhibition reduces proliferation and viability of del(5q) cells in vitro and in vivo, and that lenalidomide-resistant del(5q) MDS cells depleted of either TP53 or RUNX1 are sensitive to IGF-1R inhibition. Resistant del(5q) MDS-L cells, as well as primary MDS marrow cells, are also sensitive to targeting of IGF-1R-related dependencies in del(5q) MDS, which include the Abl and MAPK signaling pathways. This work thus provides potential new therapeutic avenues for lenalidomide-resistant del(5q) MDS.

miR-143和miR-145的单倍性不足揭示了耐药del(5q)骨髓增生异常肿瘤的靶向依赖性
骨髓增生异常肿瘤(MDS)是一种以造血功能低下和转化为急性髓性白血病(AML)的风险为特征的干细胞疾病。染色体改变在MDS中很常见,其中5q染色体间质性缺失(del(5q))最为常见。来那度胺是目前治疗del(5q) MDS的一线药物,其疗效依赖于位于5q染色体常缺失区(CDR)的CSNK1A1基因编码的CK1α的降解。然而,来那度胺耐药是常见的,通常继发于TP53或RUNX1的功能丧失突变。del(5q)中的CDR包含几个基因,包括非编码mirna,这些基因的缺失会导致疾病表型。miR-143和miR-145位于del(5q) CDR中,但对它们在人类造血和del(5q) MDS发病机制中的作用缺乏精确的了解。在这里,我们提供的证据表明,miR-143和miR-145的缺乏在del(5q) MDS的克隆扩增中起作用。我们发现胰岛素样生长因子1受体(IGF-1R)是miR-143和miR-145的直接靶点。我们的数据表明,在体外和体内,IGF-1R抑制降低了del(5q)细胞的增殖和活力,并且来那度胺耐药的del(5q) MDS细胞缺乏TP53或RUNX1对IGF-1R抑制敏感。耐药的del(5q) MDS- l细胞,以及原代MDS骨髓细胞,也对del(5q) MDS中igf - 1r相关依赖的靶向敏感,包括Abl和MAPK信号通路。因此,这项工作为来那度胺耐药del(5q) MDS提供了潜在的新治疗途径。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Leukemia
医学-血液学
CiteScore
18.10
自引率
3.50%
发文量
270
审稿时长
3-6 weeks
期刊介绍:
Title: Leukemia
Journal Overview:
Publishes high-quality, peer-reviewed research
Covers all aspects of research and treatment of leukemia and allied diseases
Includes studies of normal hemopoiesis due to comparative relevance
Topics of Interest:
Oncogenes
Growth factors
Stem cells
Leukemia genomics
Cell cycle
Signal transduction
Molecular targets for therapy
And more
Content Types:
Original research articles
Reviews
Letters
Correspondence
Comments elaborating on significant advances and covering topical issues
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


