Enantiocontrolled Cyclization to Form Chiral 7- and 8-Membered Rings Unified by the Same Catalyst Operating with Different Mechanisms
IF 14.4
1区 化学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
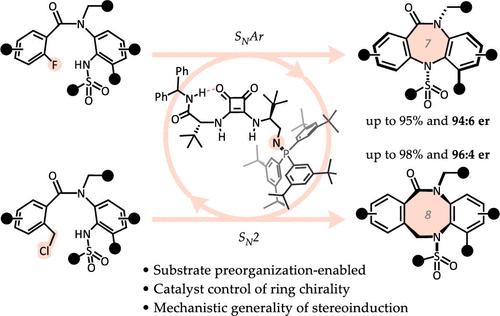
Chiral medium-sized rings, albeit displaying attractive properties for drug development, suffer from numerous synthetic challenges due to difficult cyclization steps that must take place to form these unusually strained, atropisomeric rings from sterically crowded precursors. In fact, catalytic enantioselective cyclization methods for the formation of chiral seven-membered rings are unknown, and the corresponding eight-membered variants are also sparse. In this work, we present a substrate preorganization-based, enantioselective, organocatalytic strategy to construct seven- and eight-membered rings featuring chirality that is intrinsic to the ring in the absence of singular stereogenic atoms or single bond axes of chirality. The reactions proceed under mild conditions and with high levels of stereocontrol. Notably, the same bifunctional iminophosphorane chiral catalyst orchestrates the cyclization of substrates of two different ring sizes, under two different mechanistic paradigms. We envision that the mechanistic and ring size versatility of this method could guide further applications of asymmetric catalysis to other challenging cyclization reactions.
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
24.40
自引率
6.00%
发文量
2398
审稿时长
1.6 months
期刊介绍:
The flagship journal of the American Chemical Society, known as the Journal of the American Chemical Society (JACS), has been a prestigious publication since its establishment in 1879. It holds a preeminent position in the field of chemistry and related interdisciplinary sciences. JACS is committed to disseminating cutting-edge research papers, covering a wide range of topics, and encompasses approximately 19,000 pages of Articles, Communications, and Perspectives annually. With a weekly publication frequency, JACS plays a vital role in advancing the field of chemistry by providing essential research.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


