A constitutive interferon-high immunophenotype defines response to immunotherapy in colorectal cancer
IF 48.8
1区 医学
Q1 CELL BIOLOGY
引用次数: 0
Abstract
Fewer than 50% of metastatic deficient mismatch repair (dMMR) colorectal cancer (CRC) patients respond to immune checkpoint inhibition (ICI). Identifying and expanding this patient population remains a pressing clinical need. Here, we report that an interferon-high immunophenotype locally enriched in cytotoxic lymphocytes and antigen-presenting macrophages is required for response. This immunophenotype is not exclusive to dMMR CRCs but comprises a subset of MMR proficient (pMMR) CRCs. Single-cell spatial analysis and in vitro cell co-cultures indicate that interferon-producing cytotoxic T cells induce overexpression of antigen presentation in adjacent macrophages and tumor cells, including MHC class II invariant chain CD74. dMMR CRCs expressing high levels of CD74 respond to ICI and a subset of CD74 high pMMR CRC patients show better progression free survival when treated with ICI. Therefore, CD74 abundance can identify the constitutive interferon-high immunophenotype determining clinical benefit in CRC, independently of tumor mutational burden or MMR status.
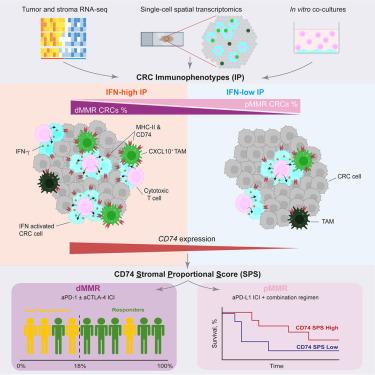
组成型干扰素高免疫表型定义了结直肠癌对免疫治疗的反应
不到50%的转移性缺陷错配修复(dMMR)结直肠癌(CRC)患者对免疫检查点抑制(ICI)有反应。确定和扩大这一患者群体仍然是迫切的临床需要。在这里,我们报告了在细胞毒性淋巴细胞和抗原呈递巨噬细胞中局部富集的干扰素高免疫表型是应答所必需的。这种免疫表型并非dMMR crc所独有,而是包括MMR精通(pMMR) crc的一个子集。单细胞空间分析和体外细胞共培养表明,产生干扰素的细胞毒性T细胞诱导邻近巨噬细胞和肿瘤细胞中抗原呈递的过表达,包括MHC II类不变链CD74。表达高水平CD74的dMMR CRC对ICI有反应,并且CD74高pMMR CRC患者的子集在接受ICI治疗时表现出更好的无进展生存期。因此,CD74丰度可以独立于肿瘤突变负担或MMR状态,识别决定结直肠癌临床获益的构成型高干扰素免疫表型。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Cancer Cell
医学-肿瘤学
CiteScore
55.20
自引率
1.20%
发文量
179
审稿时长
4-8 weeks
期刊介绍:
Cancer Cell is a journal that focuses on promoting major advances in cancer research and oncology. The primary criteria for considering manuscripts are as follows:
Major advances: Manuscripts should provide significant advancements in answering important questions related to naturally occurring cancers.
Translational research: The journal welcomes translational research, which involves the application of basic scientific findings to human health and clinical practice.
Clinical investigations: Cancer Cell is interested in publishing clinical investigations that contribute to establishing new paradigms in the treatment, diagnosis, or prevention of cancers.
Insights into cancer biology: The journal values clinical investigations that provide important insights into cancer biology beyond what has been revealed by preclinical studies.
Mechanism-based proof-of-principle studies: Cancer Cell encourages the publication of mechanism-based proof-of-principle clinical studies, which demonstrate the feasibility of a specific therapeutic approach or diagnostic test.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


