Multiple Reaction Pathways for Oxygen Evolution as a Key Factor for the Catalytic Activity of Nickel–Iron (Oxy)Hydroxides
IF 14.4
1区 化学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
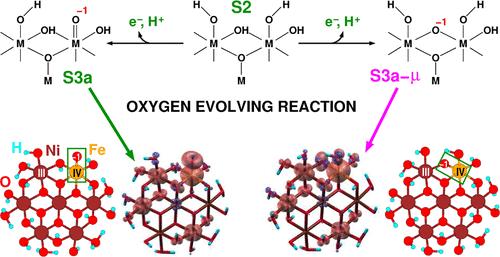
We present a comprehensive theoretical study, using state-of-the-art density functional theory simulations, of the structural and electrochemical properties of amorphous pristine and iron-doped nickel–(oxy)hydroxide catalyst films for water oxidation in alkaline solutions, referred to as NiCat and Fe:NiCat. Our simulations accurately capture the structural changes in locally ordered units, as reported by X-ray absorption spectroscopy, when the catalyst films are activated by exposure to a positive potential. We emphasize the critical role of proton-coupled electron transfer in the reversible oxidation of Ni(II) to Ni(III/IV) during this activation. After establishing the structural models of NiCat and Fe:NiCat consistent with experimental data, we used them to explore the atomistic mechanism of the oxygen evolution reaction (OER), which is triggered once the applied potential exceeds the overpotential required for water oxidation and oxygen production. We quantitatively compared seven OER pathways applicable to both the adsorbate evolution mechanism (AEM) and the lattice-oxygen-mediated mechanism (LOM) families, elucidating how iron significantly enhances the catalytic activity of Fe:NiCat compared to NiCat. Our findings suggest that simple metal–oxygen–metal motifs, common on the surface of both crystalline and amorphous metal (oxy)hydroxide films, can promote both AEM and LOM mechanisms under typical OER conditions. Furthermore, we propose that the elusive role of iron lies in the distinct behavior of Ni(IV)–O and Fe(IV)–O bonds in key intermediates preceding the formation of the O–O bond, with Fe ions lowering the potential needed to form these intermediates across the investigated pathways.
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
24.40
自引率
6.00%
发文量
2398
审稿时长
1.6 months
期刊介绍:
The flagship journal of the American Chemical Society, known as the Journal of the American Chemical Society (JACS), has been a prestigious publication since its establishment in 1879. It holds a preeminent position in the field of chemistry and related interdisciplinary sciences. JACS is committed to disseminating cutting-edge research papers, covering a wide range of topics, and encompasses approximately 19,000 pages of Articles, Communications, and Perspectives annually. With a weekly publication frequency, JACS plays a vital role in advancing the field of chemistry by providing essential research.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


