Temperature Induced Unfolding and Compaction of Cytochrome c in the Same Aqueous Solutions
IF 14.4
1区 化学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
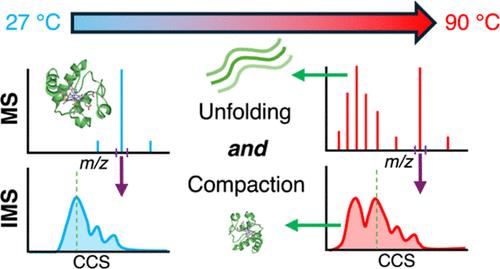
Most conventional methods used to measure protein melting temperatures reflect changes in structure between different conformational states and are typically fit to a two-state model. Population abundances of distinct conformations were measured using variable-temperature electrospray ionization ion mobility mass spectrometry to investigate the thermally induced unfolding of the model protein cytochrome c. Nineteen conformers formed at high temperature have elongated structures, consistent with unfolded forms of this protein. However, one conformer that is more compact than the native state of the protein is also formed from this same solution upon heating. The abundance of this compact conformer increases with temperatures up to 90 °C. Rapid mixing and collision-induced gas-phase unfolding experiments demonstrate that formation of this compact conformer is not an artifact of rapid refolding during the ESI process or structural rearrangement in the gas-phase, and therefore the compact conformer must be formed in bulk solution at higher temperatures. The main folded conformer at 90 °C has a cross section that is ∼30 Å2 larger than that at 27 °C. Results from collision-induced unfolding experiments indicate that they have different gas-phase stabilities that are not directly related to differences in their initial internal energies upon transitioning into the gas phase and therefore have different structures. These results demonstrate the advantage of mass and ion mobility measurements for investigating protein conformational landscapes and provide the first evidence for formation of both unfolded and more compact conformations of a protein from the same solution upon heating.
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
24.40
自引率
6.00%
发文量
2398
审稿时长
1.6 months
期刊介绍:
The flagship journal of the American Chemical Society, known as the Journal of the American Chemical Society (JACS), has been a prestigious publication since its establishment in 1879. It holds a preeminent position in the field of chemistry and related interdisciplinary sciences. JACS is committed to disseminating cutting-edge research papers, covering a wide range of topics, and encompasses approximately 19,000 pages of Articles, Communications, and Perspectives annually. With a weekly publication frequency, JACS plays a vital role in advancing the field of chemistry by providing essential research.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


