NUFIP1 integrates amino acid sensing and DNA damage response to maintain the intestinal homeostasis
IF 20.8
1区 医学
Q1 ENDOCRINOLOGY & METABOLISM
引用次数: 0
Abstract
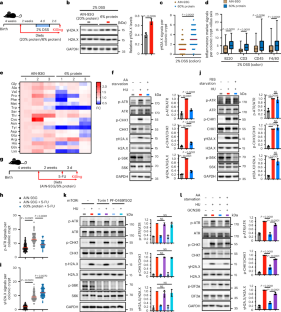
Nutrient availability strongly affects intestinal homeostasis. Here, we report that low-protein (LP) diets decrease amino acids levels, impair the DNA damage response (DDR), cause DNA damage and exacerbate inflammation in intestinal tissues of male mice with inflammatory bowel disease (IBD). Intriguingly, loss of nuclear fragile X mental retardation-interacting protein 1 (NUFIP1) contributes to the amino acid deficiency-induced impairment of the DDR in vivo and in vitro and induces necroptosis-related spontaneous enteritis. Mechanistically, phosphorylated NUFIP1 binds to replication protein A2 (RPA32) to recruit the ataxia telangiectasia and Rad3-related (ATR)–ATR-interacting protein (ATRIP) complex, triggering the DDR. Consistently, both reintroducing NUFIP1 but not its non-phospho-mutant and inhibition of necroptosis prevent bowel inflammation in male Nufip1 conditional knockout mice. Intestinal inflammation and DNA damage in male mice with IBD can be mitigated by NUFIP1 overexpression. Moreover, NUFIP1 protein levels in the intestine of patients with IBD were found to be significantly decreased. Conclusively, our study uncovers that LP diets contribute to intestinal inflammation by hijacking NUFIP1–DDR signalling and thereby activating necroptosis. Under low-protein conditions, decrease of protein NUFIP1 is found to impair DNA damage response and induce necroptosis in the intestine, thereby contributing to intestinal inflammation in male mice with inflammatory bowel disease.

NUFIP1整合了氨基酸感知和DNA损伤反应来维持肠道内稳态
营养可利用性强烈影响肠道内稳态。在这里,我们报道了低蛋白(LP)饮食降低了患有炎症性肠病(IBD)的雄性小鼠的氨基酸水平,损害了DNA损伤反应(DDR),导致DNA损伤并加剧了肠道组织的炎症。有趣的是,核脆性X智力迟钝相互作用蛋白1 (NUFIP1)的缺失导致体内和体外氨基酸缺乏诱导的DDR损伤,并诱导坏死相关的自发性肠炎。机制上,磷酸化的NUFIP1结合复制蛋白A2 (RPA32)募集共济失调毛细血管扩张和rad3相关(ATR) - ATR相互作用蛋白(ATRIP)复合物,触发DDR。同样,重新引入NUFIP1而不是其非磷酸化突变体和抑制坏死性上睑下垂都可以预防雄性NUFIP1条件敲除小鼠的肠道炎症。NUFIP1过表达可减轻IBD雄性小鼠的肠道炎症和DNA损伤。此外,发现IBD患者肠道中NUFIP1蛋白水平显著降低。最后,我们的研究发现,低脂饮食通过劫持NUFIP1-DDR信号,从而激活坏死下垂,从而促进肠道炎症。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
Nature metabolism
ENDOCRINOLOGY & METABOLISM-
CiteScore
27.50
自引率
2.40%
发文量
170
期刊介绍:
Nature Metabolism is a peer-reviewed scientific journal that covers a broad range of topics in metabolism research. It aims to advance the understanding of metabolic and homeostatic processes at a cellular and physiological level. The journal publishes research from various fields, including fundamental cell biology, basic biomedical and translational research, and integrative physiology. It focuses on how cellular metabolism affects cellular function, the physiology and homeostasis of organs and tissues, and the regulation of organismal energy homeostasis. It also investigates the molecular pathophysiology of metabolic diseases such as diabetes and obesity, as well as their treatment. Nature Metabolism follows the standards of other Nature-branded journals, with a dedicated team of professional editors, rigorous peer-review process, high standards of copy-editing and production, swift publication, and editorial independence. The journal has a high impact factor, has a certain influence in the international area, and is deeply concerned and cited by the majority of scholars.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


