Enantioselective Synthesis of Chiral β2-Amino Phosphorus Derivatives via Nickel-Catalyzed Asymmetric Hydrogenation
IF 14.4
1区 化学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
Compared with chiral β3-amino phosphorus compounds, which can be easily derived from natural optically pure α-amino acids, obtaining chiral β2-amino phosphorus derivatives remains a challenge. These derivatives, which cannot be derived from chiral natural amino acids, possess unique biological activities or potential catalytic activities. Herein, highly enantioselective hydrogenation for the preparation of chiral β2-amino phosphorus derivatives from E-β-enamido phosphorus compounds is reported by using a green and low-cost earth-abundant metal nickel catalyst (13 examples of 99% ee). In particular, this catalytic system provides the same enantiomer product from the E- and Z-alkene substrates, and the E/Z-substrate mixtures provide good results (up to 96% ee). The products can be diversely derivatized, and the derivatives exhibit good catalytic activities as novel chiral β2-aminophosphine ligands. Density functional theory calculations reveal that the weak attractive interactions between the nickel catalyst and the substrate are crucial for achieving perfect enantioselectivities. In addition, the different coordination modes between the E- or Z-substrates and the catalyst may result in the formation of the same enantiomer product.
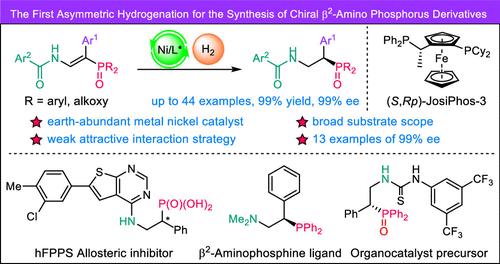
镍催化不对称氢化手性β2-氨基磷衍生物的对映选择性合成
手性β3-氨基磷化合物可以很容易地从天然光学纯α-氨基酸中得到,而获得手性β2-氨基磷衍生物仍然是一个挑战。这些衍生物具有独特的生物活性或潜在的催化活性,不能由手性天然氨基酸衍生。本文报道了利用一种绿色低成本的稀土金属镍催化剂(13例99% ee),利用E-β-enamido磷化合物进行高对映选择性加氢制备手性β2-氨基磷衍生物。特别是,该催化体系从E-烯烃和z -烯烃底物中获得相同的对映体产物,E/ z -底物混合物提供了良好的结果(高达96% ee)。产物可以多种衍生,衍生物作为新型手性β2-氨基膦配体具有良好的催化活性。密度泛函理论计算表明,镍催化剂和底物之间的弱吸引相互作用对于实现完美的对映选择性至关重要。此外,E-或z -底物与催化剂之间不同的配位模式可能导致形成相同的对映体产物。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
24.40
自引率
6.00%
发文量
2398
审稿时长
1.6 months
期刊介绍:
The flagship journal of the American Chemical Society, known as the Journal of the American Chemical Society (JACS), has been a prestigious publication since its establishment in 1879. It holds a preeminent position in the field of chemistry and related interdisciplinary sciences. JACS is committed to disseminating cutting-edge research papers, covering a wide range of topics, and encompasses approximately 19,000 pages of Articles, Communications, and Perspectives annually. With a weekly publication frequency, JACS plays a vital role in advancing the field of chemistry by providing essential research.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


