Copper-Catalyzed Cyclization and Alkene Transposition Cascade Enables a Modular Synthesis of Complex Spirocyclic Ethers
IF 14.4
1区 化学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
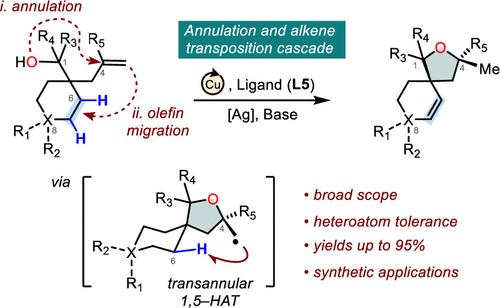
Complexity-generating reactions that access three-dimensional products from simple starting materials offer substantial value for drug discovery. While oxygen-containing heterocycles frequently feature unique, nonaromatic architectures such as spirocyclic rings, exploration of these chemical spaces is limited by conventional synthetic approaches. Herein, we report a copper-catalyzed annulation and alkene transposition cascade reaction that enables a modular preparation of complex, spirocyclic ethers from readily available alkenol substrates via a copper-catalyzed annulation and transannular 1,5-hydrogen atom transfer-mediated C–H functionalization. Our transformation displays a broad substrate scope, shows excellent heteroatom compatibility, and readily constructs spirocycles of varying ring sizes. The wider synthetic utility of this method is highlighted by numerous product diversifications and a short synthesis of the all-carbon framework of spirotenuipesine A. We anticipate that this transformation can significantly streamline access to a privileged class of three-dimensional oxygen-containing heterocycles and will find broad application in natural product synthesis.
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
24.40
自引率
6.00%
发文量
2398
审稿时长
1.6 months
期刊介绍:
The flagship journal of the American Chemical Society, known as the Journal of the American Chemical Society (JACS), has been a prestigious publication since its establishment in 1879. It holds a preeminent position in the field of chemistry and related interdisciplinary sciences. JACS is committed to disseminating cutting-edge research papers, covering a wide range of topics, and encompasses approximately 19,000 pages of Articles, Communications, and Perspectives annually. With a weekly publication frequency, JACS plays a vital role in advancing the field of chemistry by providing essential research.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


