Daily glucocorticoids promote glioblastoma growth and circadian synchrony to the host
IF 48.8
1区 医学
Q1 CELL BIOLOGY
引用次数: 0
Abstract
Glioblastoma (GBM) is the most common primary malignant brain tumor in adults with a poor prognosis despite aggressive therapy. Here, we hypothesized that daily host signaling regulates tumor growth and synchronizes circadian rhythms in GBM. We find daily glucocorticoids promote or suppress GBM growth through glucocorticoid receptor (GR) signaling depending on time of day and the clock genes, Bmal1 and Cry. Blocking circadian signals, like vasoactive intestinal peptide or glucocorticoids, dramatically slows GBM growth and disease progression. Analysis of human GBM samples from The Cancer Genome Atlas (TCGA) shows that high GR expression significantly increases hazard of mortality. Finally, mouse and human GBM models have intrinsic circadian rhythms in clock gene expression in vitro and in vivo that entrain to the host through glucocorticoid signaling, regardless of tumor type or host immune status. We conclude that GBM entrains to the circadian circuit of the brain, modulating its growth through clock-controlled cues, like glucocorticoids.
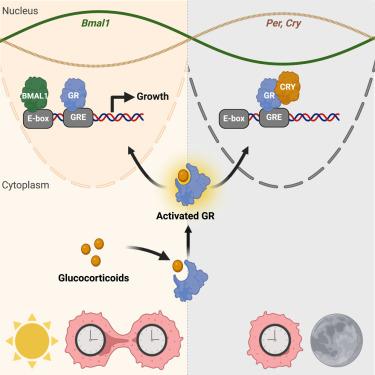
每日糖皮质激素促进胶质母细胞瘤的生长和宿主的昼夜节律同步
胶质母细胞瘤(GBM)是成人最常见的原发性恶性脑肿瘤,尽管积极治疗,预后却很差。在这里,我们假设每日宿主信号调节肿瘤生长并同步GBM的昼夜节律。我们发现每日糖皮质激素通过糖皮质激素受体(GR)信号传导促进或抑制GBM生长,这取决于一天中的时间和时钟基因Bmal1和Cry。阻断昼夜节律信号,如血管活性肠肽或糖皮质激素,可显著减缓GBM的生长和疾病进展。来自癌症基因组图谱(TCGA)的人类GBM样本分析显示,高GR表达显著增加死亡风险。最后,小鼠和人类GBM模型在体外和体内具有内在的生物钟基因表达昼夜节律,无论肿瘤类型或宿主免疫状态如何,都可以通过糖皮质激素信号传递到宿主。我们得出的结论是,GBM与大脑的昼夜节律回路有关,通过糖皮质激素等生物钟控制的线索调节其生长。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Cancer Cell
医学-肿瘤学
CiteScore
55.20
自引率
1.20%
发文量
179
审稿时长
4-8 weeks
期刊介绍:
Cancer Cell is a journal that focuses on promoting major advances in cancer research and oncology. The primary criteria for considering manuscripts are as follows:
Major advances: Manuscripts should provide significant advancements in answering important questions related to naturally occurring cancers.
Translational research: The journal welcomes translational research, which involves the application of basic scientific findings to human health and clinical practice.
Clinical investigations: Cancer Cell is interested in publishing clinical investigations that contribute to establishing new paradigms in the treatment, diagnosis, or prevention of cancers.
Insights into cancer biology: The journal values clinical investigations that provide important insights into cancer biology beyond what has been revealed by preclinical studies.
Mechanism-based proof-of-principle studies: Cancer Cell encourages the publication of mechanism-based proof-of-principle clinical studies, which demonstrate the feasibility of a specific therapeutic approach or diagnostic test.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


