CTCF enhances pancreatic cancer progression via FLG-AS1-dependent epigenetic regulation and macrophage polarization
IF 15.4
1区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
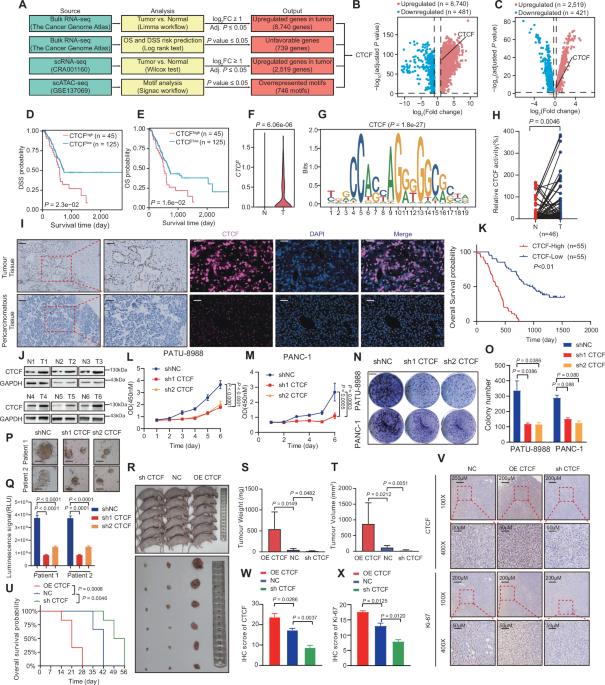
CCCTC-binding factor (CTCF) regulates chromatin organization and is upregulated in pancreatic ductal adenocarcinoma (PDAC). We found that CTCF interacts with HNRNPU through a FLG-AS1-dependent mechanism, facilitating the recruitment of EP300 and activation of the m6A reader IGF2BP2. This activation promotes histone lactylation at the promoter region of IGF2BP2 stimulating the proliferation of PDAC cells. IGF2BP2 enhanced the mRNA stability of CSF1 and MYC. Moreover, FLG-AS1 directly interacts with HNRNPU to modulate alternative splicing of CSF1, thus promoting the M2 polarization of tumor associated macrophages (TAMs) in PDAC. The results indicated that CTCF-induced oncogenic modification of histone lactylation, m6A and alternative spilcing as multi-regulation modes of TAMs reprogramming in PDAC and identifies CTCF as a potential therapeutic target for PDAC immunotherapy whose inhibition M2 polarization through the IGF2BP2/CSF1/CSF1R axis. Curaxin combined with gemcitabine treatment has shown promising antitumor efficacy against PDAC.
CTCF通过flg - as1依赖性表观遗传调控和巨噬细胞极化促进胰腺癌进展
ccctc结合因子(CTCF)调节染色质组织,并在胰腺导管腺癌(PDAC)中上调。我们发现CTCF通过flg - as1依赖机制与HNRNPU相互作用,促进EP300的募集和m6A读取器IGF2BP2的激活。这种激活促进IGF2BP2启动子区域的组蛋白乳酸化,刺激PDAC细胞的增殖。IGF2BP2增强了CSF1和MYC mRNA的稳定性。此外,FLG-AS1直接与HNRNPU相互作用,调节CSF1的选择性剪接,从而促进PDAC中肿瘤相关巨噬细胞(tumor associated macrophages, tam)的M2极化。结果表明,CTCF诱导组蛋白乙酰化、m6A和选择性溢出的致癌修饰是PDAC中tam重编程的多调控模式,并确定CTCF是PDAC免疫治疗的潜在治疗靶点,其通过IGF2BP2/CSF1/CSF1R轴抑制M2极化。Curaxin联合吉西他滨治疗PDAC显示出良好的抗肿瘤效果。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Cell Death and Differentiation
生物-生化与分子生物学
CiteScore
24.70
自引率
1.60%
发文量
181
审稿时长
3 months
期刊介绍:
Mission, vision and values of Cell Death & Differentiation:
To devote itself to scientific excellence in the field of cell biology, molecular biology, and biochemistry of cell death and disease.
To provide a unified forum for scientists and clinical researchers
It is committed to the rapid publication of high quality original papers relating to these subjects, together with topical, usually solicited, reviews, meeting reports, editorial correspondence and occasional commentaries on controversial and scientifically informative issues.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


