Anticancer drug design of potassium ion (K+) assisted self-assembled Cu(II) Schiff base complex and its Ni(II) and Zn(II) analogues (3d-4s): A comparative study of DNA/protein (BSA) interactions and cytotoxicity
IF 4
2区 化学
Q2 CHEMISTRY, PHYSICAL
引用次数: 0
Abstract
In the research for the therapeutic agents, three novel dinuclear 3d-4s complexes with a glycine-derived Schiff base ligand and a side-on bridging azido (µ-1,3-N3) ligand were synthesized and characterized, with formulas [K(CuL)(µ-1,3-N3)] (KCuL), [K(NiL)(µ-1,3-N3)] (KNiL) and [K(ZnL)(µ-1,3-N3)] (KZnL), where L indicates the Schiff base containing stoichiometric ratio of glycine and salicylaldehyde. Different structural, spectral and analytical methods, such as single-crystal X-ray crystallography, CHN analysis, FT-IR, UV–Vis and mass spectroscopy, were used for the characterization. The single-crystal X-ray diffraction of the KCuL complex confirmed the presence of a dinuclear complex containing one Cu(II) and K+centre, one Schiff base ligand and one side-on exogenous bridging azido ligand (μ-1,3-N3) in the asymmetric unit. A considerable anticancer activity of the complexes was explored on different cancerous cell lines such as breast, lung carcinoma, gastric and enocarcinoma and normal fibroblasts. DNA cleavage experiment showed that all the complexes cleave pGFPC1 supercoiled DNA in the presence of an activator like H2O2. The interaction between the complexes and Fish Sperm DNA (FS-DNA) was inspected by UV–Vis and fluorescence titration spectrometry, viscosity measurement, cyclic voltammetry and docking studies, with intercalation found as the main binding mode. The order of intercalation propensity of the complexes was deduced from Kb values as KCuL > KNiL > KZnL, and this order was confirmed by the competitive EtBr/complex binding experiment. Absorption and fluorescence spectroscopy revealed a static quenching mechanism as the interaction mechanism for binding between the complexes and bovine serum albumin protein (BSA). Synchronous fluorescence and site-marker competitive experiments completed the investigations. Also, Forster resonance energy transfer theory (FRET) was employed to calculate the distance, r, between the acceptor (complex) and the donor (BSA). The quantitative tendency of the complexes to interact with BSA was calculated as KSV, which varied in the order KCuL > KNiL > KZnL. Besides the results from the experimental methods, the theoretical molecular docking study and the Hirshfeld surface study alongside finger print plots were applied to confirm the mode and site of binding of the KCuL complex with DNA and BSA.
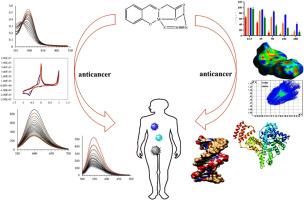
钾离子(K+)辅助自组装Cu(II)希夫碱配合物及其Ni(II)和Zn(II)类似物的抗癌药物设计(3d-4s): DNA/蛋白(BSA)相互作用和细胞毒性的比较研究
在治疗药物的研究中,合成了甘氨酸衍生的希夫碱配体和侧接叠氮(µ-1,3- n3)配体的三种新型双核3d-4s配合物,并对其进行了表征,其分子式为[K(CuL)(µ-1,3- n3)] (KCuL)、[K(NiL)(µ-1,3- n3)] (KNiL)和[K(ZnL)(µ-1,3- n3)] (KZnL),其中L表示希夫碱中甘氨酸和水杨醛的化学计量比。采用不同的结构、光谱和分析方法,如单晶x射线晶体学、CHN分析、FT-IR、UV-Vis和质谱等对其进行表征。KCuL配合物的单晶x射线衍射证实了在不对称单元中存在一个双核配合物,包含一个Cu(II)和K+中心,一个席夫碱配体和一个外源桥接叠氮配体(μ-1,3- n3)。该复合物对乳腺癌、肺癌、胃癌、肠腺癌及正常成纤维细胞等不同类型的癌细胞均有明显的抗癌活性。DNA裂解实验表明,在H2O2等激活剂存在的情况下,所有复合物都能裂解pGFPC1超螺旋DNA。采用UV-Vis、荧光滴定法、粘度测定、循环伏安法和对接研究等方法考察了配合物与鱼精子DNA (FS-DNA)的相互作用,发现嵌入层是配合物的主要结合方式。根据Kb值推导出配合物的插层倾向顺序为:KCuL >;KNiL祝辞KZnL,这个顺序被竞争性的EtBr/配合物结合实验证实。吸收光谱和荧光光谱分析表明,配合物与牛血清白蛋白(BSA)结合的相互作用机制为静态猝灭机制。同步荧光和位点标记竞争实验完成了研究。利用福斯特共振能量转移理论(FRET)计算了受体(配合物)与供体(BSA)之间的距离r。配合物与BSA相互作用的定量趋势计算为KSV,其变化顺序为KCuL >;KNiL祝辞KZnL。除了实验方法的结果外,我们还利用理论分子对接研究和Hirshfeld表面研究以及指纹图来确定KCuL复合物与DNA和BSA结合的方式和位点。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
Journal of Molecular Structure
化学-物理化学
CiteScore
7.10
自引率
15.80%
发文量
2384
审稿时长
45 days
期刊介绍:
The Journal of Molecular Structure is dedicated to the publication of full-length articles and review papers, providing important new structural information on all types of chemical species including:
• Stable and unstable molecules in all types of environments (vapour, molecular beam, liquid, solution, liquid crystal, solid state, matrix-isolated, surface-absorbed etc.)
• Chemical intermediates
• Molecules in excited states
• Biological molecules
• Polymers.
The methods used may include any combination of spectroscopic and non-spectroscopic techniques, for example:
• Infrared spectroscopy (mid, far, near)
• Raman spectroscopy and non-linear Raman methods (CARS, etc.)
• Electronic absorption spectroscopy
• Optical rotatory dispersion and circular dichroism
• Fluorescence and phosphorescence techniques
• Electron spectroscopies (PES, XPS), EXAFS, etc.
• Microwave spectroscopy
• Electron diffraction
• NMR and ESR spectroscopies
• Mössbauer spectroscopy
• X-ray crystallography
• Charge Density Analyses
• Computational Studies (supplementing experimental methods)
We encourage publications combining theoretical and experimental approaches. The structural insights gained by the studies should be correlated with the properties, activity and/ or reactivity of the molecule under investigation and the relevance of this molecule and its implications should be discussed.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


