Evaluation of isatin-1,3,5-triazine-benzylamine hybrids as multi-target directed ligands against Alzheimer's disease
IF 4
2区 化学
Q2 CHEMISTRY, PHYSICAL
引用次数: 0
Abstract
In this study a new series of isatin-1,3,5-triazine-benzylamine hybrids (6a-k) were designed, synthesized and evaluated for in vitro anti-Alzheimer's disease(AD) properties. These molecules were tested for acetylcholinesterase (AChE) and butyrylcholinesterase (BChE) enzymes inhibition and biometal chelation effect. Results revealed these molecules to be very potent AChE inhibitors with IC50 values in the range of 0.86–36.30 nM. BChE inhibition potencies were also good and IC50 values for active compounds were from 0.66 to 3.88 μM. Molecule 6c had the highest activity against both enzymes and further studies showed that it kinetically inhibited AChE and BChE with non-competitive and competitive mechanisms, respectively. This compound could effectively inhibit amyloid beta self/Cu2+ induced aggregation in 10μM. These compounds could significantly chelate to Cu2+ as an important biometal involved in AD andin silico simulations demonstrated that these molecules had key interactions with both ChE enzymes. It was concluded that these compounds had high potency to be used in future evaluations as anti-AD lead compounds.
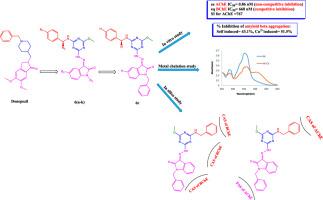
isatin-1,3,5-三嗪-苄胺杂合体作为多靶点定向配体治疗阿尔茨海默病的评价
本研究设计、合成了一系列新的isatin-1,3,5-三嗪-苄胺杂合体(6a-k),并对其体外抗阿尔茨海默病(AD)的性能进行了评价。检测了这些分子对乙酰胆碱酯酶(AChE)和丁基胆碱酯酶(BChE)的抑制作用和生物金属螯合作用。结果表明,这些分子是非常有效的AChE抑制剂,IC50值在0.86 ~ 36.30 nM范围内。对BChE的抑制作用也较好,活性化合物的IC50值在0.66 ~ 3.88 μM之间。分子6c对这两种酶的活性最高,进一步的研究表明,它分别通过非竞争和竞争机制抑制AChE和BChE。该化合物能有效抑制β淀粉样蛋白自身/Cu2+在10μM内诱导的聚集。这些化合物可以明显螯合Cu2+作为AD的重要生物金属,并且硅模拟表明这些分子与两种ChE酶具有关键的相互作用。这些化合物作为抗阿尔茨海默病的先导化合物具有较高的效价。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
Journal of Molecular Structure
化学-物理化学
CiteScore
7.10
自引率
15.80%
发文量
2384
审稿时长
45 days
期刊介绍:
The Journal of Molecular Structure is dedicated to the publication of full-length articles and review papers, providing important new structural information on all types of chemical species including:
• Stable and unstable molecules in all types of environments (vapour, molecular beam, liquid, solution, liquid crystal, solid state, matrix-isolated, surface-absorbed etc.)
• Chemical intermediates
• Molecules in excited states
• Biological molecules
• Polymers.
The methods used may include any combination of spectroscopic and non-spectroscopic techniques, for example:
• Infrared spectroscopy (mid, far, near)
• Raman spectroscopy and non-linear Raman methods (CARS, etc.)
• Electronic absorption spectroscopy
• Optical rotatory dispersion and circular dichroism
• Fluorescence and phosphorescence techniques
• Electron spectroscopies (PES, XPS), EXAFS, etc.
• Microwave spectroscopy
• Electron diffraction
• NMR and ESR spectroscopies
• Mössbauer spectroscopy
• X-ray crystallography
• Charge Density Analyses
• Computational Studies (supplementing experimental methods)
We encourage publications combining theoretical and experimental approaches. The structural insights gained by the studies should be correlated with the properties, activity and/ or reactivity of the molecule under investigation and the relevance of this molecule and its implications should be discussed.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


