Elucidating acquired PARP inhibitor resistance in advanced prostate cancer
IF 48.8
1区 医学
Q1 CELL BIOLOGY
引用次数: 0
Abstract
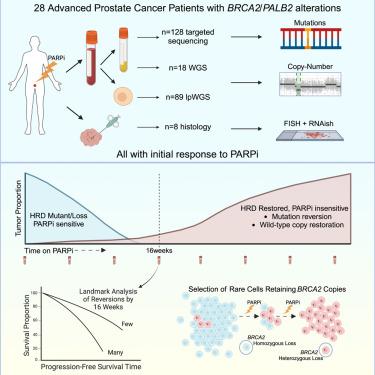
PARP inhibition (PARPi) has anti-tumor activity against castration-resistant prostate cancer (CRPC) with homologous recombination repair (HRR) defects. However, mechanisms underlying PARPi resistance are not fully understood. While acquired mutations restoring BRCA genes are well documented, their clinical relevance, frequency, and mechanism of generation remain unclear. Moreover, how resistance emerges in BRCA2 homozygously deleted (HomDel) CRPC is unknown. Evaluating samples from patients with metastatic CRPC treated in the TOPARP-B trial, we identify reversion mutations in most BRCA2/PALB2-mutated tumors (79%) by end of treatment. Among reversions mediated by frameshift deletions, 60% are flanked by DNA microhomologies, implicating POLQ-mediated repair. The number of reversions and time of their detection associate with radiological progression-free survival and overall survival (p < 0.01). For BRCA2 HomDels, selection for rare subclones without BRCA2-HomDel is observed following PARPi, confirmed by single circulating-tumor-cell genomics, biopsy fluorescence in situ hybridization (FISH), and RNAish. These data support the need for restored HRR function in PARPi resistance.
阐明晚期前列腺癌获得性 PARP 抑制剂耐药性
PARP 抑制剂(PARPi)对存在同源重组修复(HRR)缺陷的去势抵抗性前列腺癌(CRPC)具有抗肿瘤活性。然而,PARPi 的耐药性机制尚未完全明了。虽然恢复 BRCA 基因的获得性突变已被充分记录,但其临床相关性、频率和产生机制仍不清楚。此外,BRCA2同源染色体缺失(HomDel)CRPC的耐药性是如何产生的也不清楚。通过评估在 TOPARP-B 试验中接受治疗的转移性 CRPC 患者样本,我们发现大多数 BRCA2/PALB2 突变肿瘤(79%)在治疗结束时出现了逆转突变。在由框移缺失介导的逆转突变中,60%的逆转突变两侧存在DNA微缺失,这与POLQ介导的修复有关。逆转的数量及其检测时间与放射学无进展生存期和总生存期相关(p < 0.01)。对于 BRCA2 HomDel,在 PARPi 之后会观察到对没有 BRCA2-HomDel 的罕见亚克隆的选择,单个循环肿瘤细胞基因组学、活检荧光原位杂交(FISH)和 RNAish 证实了这一点。这些数据支持了在 PARPi 抗性中恢复 HRR 功能的必要性。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Cancer Cell
医学-肿瘤学
CiteScore
55.20
自引率
1.20%
发文量
179
审稿时长
4-8 weeks
期刊介绍:
Cancer Cell is a journal that focuses on promoting major advances in cancer research and oncology. The primary criteria for considering manuscripts are as follows:
Major advances: Manuscripts should provide significant advancements in answering important questions related to naturally occurring cancers.
Translational research: The journal welcomes translational research, which involves the application of basic scientific findings to human health and clinical practice.
Clinical investigations: Cancer Cell is interested in publishing clinical investigations that contribute to establishing new paradigms in the treatment, diagnosis, or prevention of cancers.
Insights into cancer biology: The journal values clinical investigations that provide important insights into cancer biology beyond what has been revealed by preclinical studies.
Mechanism-based proof-of-principle studies: Cancer Cell encourages the publication of mechanism-based proof-of-principle clinical studies, which demonstrate the feasibility of a specific therapeutic approach or diagnostic test.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


