Methodological and reporting quality of systematic and rapid reviews on human mpox and their utility during a public health emergency
Abstract
Introduction
Evidence syntheses were rapidly produced during the 2022 mpox outbreak despite a lack of studies. The aim of this methodological study was to assess the quality and utility of the evidence syntheses produced during the first 6 months of the outbreak compared to those published before it.
Methods
Human mpox evidence syntheses available before December 31, 2022 were retrieved from PubMed, Scopus, EuropePMC, SSRN, and arXiv. Study characteristics, utility, methodological, and reporting quality (AMSTAR-2 and PRISMA) were contrasted between syntheses produced before the 2022 outbreak (historical) and during the first 6 months (new). Results were synthesized narratively.
Results
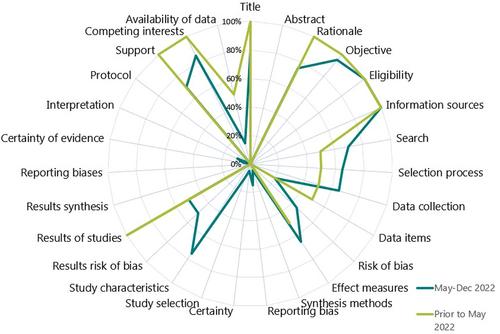
Twenty-six evidence syntheses were included; two historical systematic reviews (SRs) and 24 new SRs, rapid reviews, scoping reviews, and mislabelled syntheses. Median time from search to publication/preprint post date was 68 and 6 weeks for historical and new syntheses, respectively. Among the new syntheses, 8% (2/24) did not include evidence from the 2022 outbreak, 33% (8/24) included only new evidence and 58% (14/24) included both new and historical evidence. Only 29% of new syntheses contrasted findings between new and historical evidence. Methodological quality was critically low for 100% of historical syntheses and 92% of new syntheses and the remainder (8%) were low. Reporting quality was poor with a median of 10.5 (range 10–11) and 11.5 (range 4–21) of 27 items reported sufficiently by historical and new syntheses, respectively.
Conclusions
Evidence syntheses take time to produce and during an emergent outbreak they are often outdated at the time of publication and suffer from poor adherence to methodological and reporting guidelines. Overlapping content and few new studies resulted in minimal added value to the mpox literature. Strategies to reduce duplication and mechanisms to produce and disseminate continuously updated living evidence syntheses need to be explored to support decision-makers responding to an emergency.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


