Structures and pH-dependent dimerization of the sevenless receptor tyrosine kinase
IF 14.5
1区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
Sevenless (Sev) is a Drosophila receptor tyrosine kinase (RTK) required for the specification of the R7 photoreceptor. It is cleaved into α and β subunits and binds the ectodomain of the G-protein-coupled receptor bride of sevenless (Boss). Previous work showed that the Boss ectodomain could bind but not activate Sev; rather, the whole seven-pass transmembrane Boss was required. Here, we show that Sev does not need to be cleaved to function and that a single-pass transmembrane form of Boss activates Sev. We use cryo-electron microscopy and biophysical methods to determine the structural basis of ligand binding and pH-dependent dimerization of Sev, and we discuss the implications in the process of Sev activation. The Sev human homolog, receptor oncogene from sarcoma 1 (ROS1), is associated with oncogenic transformations, and we discuss their structural similarities.
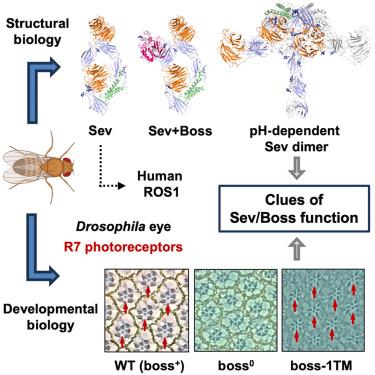
无七受体酪氨酸激酶的结构和 pH 依赖性二聚化
七无(Sev)是果蝇受体酪氨酸激酶(RTK),R7感光器的规格化需要它。它被裂解成 α 和 β 亚基,并与七无 G 蛋白偶联受体新娘(Boss)的外显子结合。以前的研究表明,Boss 外结构域能与 Sev 结合,但不能激活 Sev;相反,需要整个七孔跨膜 Boss。在这里,我们证明 Sev 不需要被裂解就能发挥作用,而且单通跨膜形式的 Boss 能激活 Sev。我们使用冷冻电镜和生物物理方法确定了配体结合和 Sev 的 pH 依赖性二聚化的结构基础,并讨论了 Sev 激活过程中的影响。Sev 的人类同源物肉瘤受体癌基因 1(ROS1)与致癌转化有关,我们讨论了它们在结构上的相似性。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Molecular Cell
生物-生化与分子生物学
CiteScore
26.00
自引率
3.80%
发文量
389
审稿时长
1 months
期刊介绍:
Molecular Cell is a companion to Cell, the leading journal of biology and the highest-impact journal in the world. Launched in December 1997 and published monthly. Molecular Cell is dedicated to publishing cutting-edge research in molecular biology, focusing on fundamental cellular processes. The journal encompasses a wide range of topics, including DNA replication, recombination, and repair; Chromatin biology and genome organization; Transcription; RNA processing and decay; Non-coding RNA function; Translation; Protein folding, modification, and quality control; Signal transduction pathways; Cell cycle and checkpoints; Cell death; Autophagy; Metabolism.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


