Osteoprotegerin secretion and its inhibition by RANKL in osteoblastic cells visualized using bioluminescence imaging
IF 3.5
2区 医学
Q2 ENDOCRINOLOGY & METABOLISM
引用次数: 0
Abstract
Bone remodeling is regulated by the interaction between receptor activator of nuclear factor kappa-B ligand (RANKL) and its receptor RANK on osteoblasts and osteoclasts, respectively. Osteoprotegerin (OPG) is secreted from osteoblasts and inhibits osteoclast differentiation by acting as a decoy receptor for RANKL. Despite its importance, the mechanism underlying the secretion of OPG remains poorly understood. Here, we applied a method of video-rate bioluminescence imaging using a fusion protein with Gaussia luciferase (GLase) and visualized the secretion of OPG from living mouse osteoblastic MC3T3-E1 cells. The bioluminescence imaging revealed that the secretion of OPG fused to GLase (OPG-GLase) occurred frequently and widely across the cell surface. Notably, co-expression of RANKL significantly reduced the secretion of OPG-GLase, indicating an inhibitory role of RANKL on OPG secretion within cells. Further imaging and biochemical analyses using deletion mutants of OPG and RANKL, as well as RANKL mutants that cause autosomal recessive osteopetrosis, demonstrated the essential role of protein-protein interaction between OPG and RANKL in the inhibition of OPG secretion. Treatment with proteasome inhibitors resulted in increased levels of OPG in both culture medium and cell lysates. However, the fold-increase of OPG was similar regardless of the presence or absence of RANKL, suggesting that the regulation of OPG secretion by RANKL is independent of proteasome activity. This report visualized the secretion of OPG from living cells and provided evidence for a novel intracellular inhibitory effect of RANKL on OPG secretion.
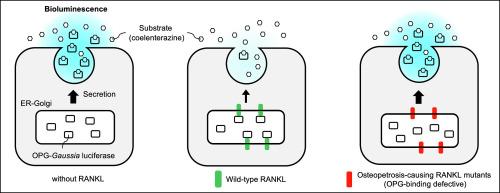
利用生物发光成像技术观察成骨细胞的骨蛋白激酶分泌及其受 RANKL 抑制的情况。
骨重塑是由成骨细胞和破骨细胞上的核因子卡巴-B 配体受体激活剂(RANKL)及其受体 RANK 之间的相互作用调节的。骨保护素(OPG)由成骨细胞分泌,通过充当 RANKL 的诱饵受体来抑制破骨细胞的分化。尽管 OPG 非常重要,但人们对其分泌机制仍然知之甚少。在这里,我们利用一种与高斯荧光素酶(GLase)融合的视频速率生物发光成像方法,观察了活体小鼠成骨细胞 MC3T3-E1 中 OPG 的分泌情况。生物发光成像显示,与高斯荧光素酶(GLase)融合的 OPG(OPG-GLase)在细胞表面频繁而广泛地分泌。值得注意的是,联合表达 RANKL 能显著减少 OPG-GLase 的分泌,这表明 RANKL 对细胞内 OPG 的分泌有抑制作用。利用 OPG 和 RANKL 的缺失突变体以及导致常染色体隐性骨质疏松症的 RANKL 突变体进行的进一步成像和生化分析表明,OPG 和 RANKL 之间的蛋白质相互作用在抑制 OPG 分泌中起着至关重要的作用。蛋白酶体抑制剂会导致培养基和细胞裂解液中的 OPG 水平升高。然而,无论是否存在 RANKL,OPG 的增加倍数都是相似的,这表明 RANKL 对 OPG 分泌的调控与蛋白酶体活性无关。该报告将活体细胞分泌 OPG 的过程可视化,并为 RANKL 对 OPG 分泌的新型细胞内抑制作用提供了证据。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
Bone
医学-内分泌学与代谢
CiteScore
8.90
自引率
4.90%
发文量
264
审稿时长
30 days
期刊介绍:
BONE is an interdisciplinary forum for the rapid publication of original articles and reviews on basic, translational, and clinical aspects of bone and mineral metabolism. The Journal also encourages submissions related to interactions of bone with other organ systems, including cartilage, endocrine, muscle, fat, neural, vascular, gastrointestinal, hematopoietic, and immune systems. Particular attention is placed on the application of experimental studies to clinical practice.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


