Targeting GSDME-mediated macrophage polarization for enhanced antitumor immunity in hepatocellular carcinoma
IF 19.8
1区 医学
Q1 IMMUNOLOGY
引用次数: 0
Abstract
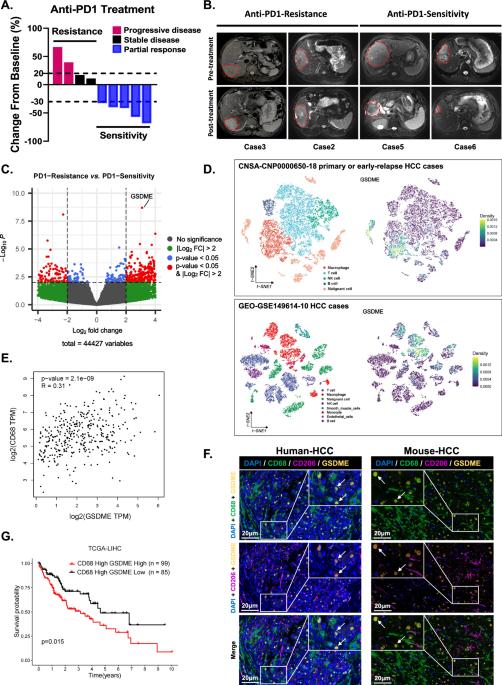
Despite the notable efficacy of anti-PD1 therapy in the management of hepatocellular carcinoma (HCC) patients, resistance in most individuals necessitates additional investigation. For this study, we collected tumor tissues from nine HCC patients receiving anti-PD1 monotherapy and conducted RNA sequencing. These findings revealed significant upregulation of GSDME, which is predominantly expressed by tumor-associated macrophages (TAMs), in anti-PD1-resistant patients. Furthermore, patients with elevated levels of GSDME+ macrophages in HCC tissues presented a poorer prognosis. The analysis of single-cell sequencing data and flow cytometry revealed that the suppression of GSDME expression in nontumor cells resulted in a decrease in the proportion of M2-like macrophages within the tumor microenvironment (TIME) of HCC while concurrently augmenting the cytotoxicity of CD8 + T cells. The non-N-terminal fragment of GSDME within macrophages combines with PDPK1, thereby activating the PI3K-AKT pathway and facilitating M2-like polarization. The small-molecule Eliprodil inhibited the increase in PDPK1 phosphorylation mediated by GSDME site 1. The combination of Eliprodil and anti-PD1 was effective in the treatment of both spontaneous HCC in c-Myc + /+;Alb-Cre + /+ mice and in a hydrodynamic tail vein injection model, which provides a promising strategy for novel combined immunotherapy.
靶向 GSDME 介导的巨噬细胞极化,增强肝细胞癌的抗肿瘤免疫力。
尽管抗-PD1疗法在治疗肝细胞癌(HCC)患者方面疗效显著,但大多数患者的耐药性仍需要进一步研究。在这项研究中,我们收集了九名接受抗 PD1 单药治疗的 HCC 患者的肿瘤组织,并进行了 RNA 测序。研究结果显示,抗PD1耐药患者体内主要由肿瘤相关巨噬细胞(TAMs)表达的GSDME明显上调。此外,HCC 组织中 GSDME+ 巨噬细胞水平升高的患者预后较差。对单细胞测序数据和流式细胞术的分析表明,抑制非肿瘤细胞中 GSDME 的表达会降低 HCC 肿瘤微环境(TIME)中 M2 样巨噬细胞的比例,同时增强 CD8 + T 细胞的细胞毒性。巨噬细胞内 GSDME 的非 N 端片段与 PDPK1 结合,从而激活 PI3K-AKT 通路并促进 M2 样极化。小分子 Eliprodil 可抑制由 GSDME 位点 1 介导的 PDPK1 磷酸化的增加。Eliprodil和抗PD1的联合疗法对c-Myc + /+;Alb-Cre + /+小鼠自发性HCC和水动力尾静脉注射模型均有效,这为新型联合免疫疗法提供了一种前景广阔的策略。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
31.20
自引率
1.20%
发文量
903
审稿时长
1 months
期刊介绍:
Cellular & Molecular Immunology, a monthly journal from the Chinese Society of Immunology and the University of Science and Technology of China, serves as a comprehensive platform covering both basic immunology research and clinical applications. The journal publishes a variety of article types, including Articles, Review Articles, Mini Reviews, and Short Communications, focusing on diverse aspects of cellular and molecular immunology.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


