PIEZO1 mediates matrix stiffness-induced tumor progression in kidney renal clear cell carcinoma by activating the Ca2+/Calpain/YAP pathway
IF 4.6
2区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
Biochimica et biophysica acta. Molecular cell research
Pub Date : 2024-10-28
DOI:10.1016/j.bbamcr.2024.119871
引用次数: 0
Abstract
Objective
The significance of physical factors in the onset and progression of tumors has been increasingly substantiated by a multitude of studies. The extracellular matrix, a pivotal component of the tumor microenvironment, has been the subject of extensive investigation in connection with the advancement of KIRC (Kidney Renal Clear Cell Carcinoma) in recent years. PIEZO1, a mechanosensitive ion channel, has been recognized as a modulator of diverse physiological processes. Nonetheless, the precise function of PIEZO1 as a transducer of mechanical stimuli in KIRC remains poorly elucidated.
Methods
A bioinformatics analysis was conducted using data from The Cancer Genome Atlas (TCGA) and the Clinical Proteomic Tumor Analysis Consortium (CPTAC) to explore the correlation between matrix stiffness indicators, such as COL1A1 and LOX mRNA levels, and KIRC prognosis. Expression patterns of mechanosensitive ion channels, particularly PIEZO1, were examined. Collagen-coated polyacrylamide hydrogel models were utilized to simulate varying stiffness environments and study their effects on KIRC cell behavior in vitro. Functional experiments, including PIEZO1 knockdown and overexpression, were performed to investigate the molecular mechanisms underlying matrix stiffness-induced cellular changes. Interventions in the Ca2+/Calpain/YAP Pathway were conducted to evaluate their effects on cell growth, EMT, and stemness characteristics.
Results
Our findings indicate a significant correlation between matrix stiffness and the prognosis of KIRC patients. It is observed that higher mechanical stiffness can facilitate the growth and metastasis of KIRC cells. Notably, we have also observed that the deficiency of PIEZO1 hinders the proliferation, EMT, and stemness characteristics of KIRC cells induced by a stiff matrix. Our study suggests that PIEZO1 plays a crucial role in mediating KIRC growth and metastasis through the activation of the Ca2+/Calpain/YAP Pathway.
Conclusion
This study elucidates a novel mechanism through which the activation of PIEZO1 leads to calcium influx, subsequent calpain activation, and YAP nuclear translocation, thereby contributing to the progression of KIRC driven by matrix stiffness.
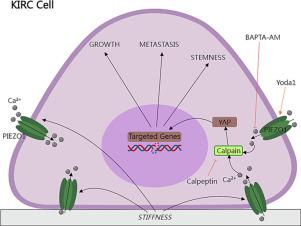
PIEZO1 通过激活 Ca2+/Calpain/YAP 通路介导基质僵化诱导的肾透明细胞癌肿瘤进展
摘要:物理因素在肿瘤发生和发展过程中的重要性已被越来越多的研究证实。细胞外基质是肿瘤微环境的重要组成部分,近年来,随着 KIRC(肾透明细胞癌)的发展,细胞外基质已成为广泛研究的主题。PIEZO1 是一种机械敏感性离子通道,被认为是多种生理过程的调节器。方法利用癌症基因组图谱(The Cancer Genome Atlas,TCGA)和临床肿瘤蛋白质组分析联盟(Clinical Proteomic Tumor Analysis Consortium,CPTAC)的数据进行生物信息学分析,探讨基质硬度指标(如COL1A1和LOX mRNA水平)与KIRC预后之间的相关性。研究还考察了机械敏感性离子通道(尤其是 PIEZO1)的表达模式。利用胶原包裹的聚丙烯酰胺水凝胶模型模拟了不同的硬度环境,并研究了它们对体外 KIRC 细胞行为的影响。进行了包括 PIEZO1 基因敲除和过表达在内的功能实验,以研究基质硬度诱导细胞变化的分子机制。我们对 Ca2+/Calpain/YAP 通路进行了干预,以评估它们对细胞生长、EMT 和干性特征的影响。我们观察到,较高的机械硬度可促进 KIRC 细胞的生长和转移。值得注意的是,我们还观察到,缺乏 PIEZO1 会阻碍僵硬基质诱导的 KIRC 细胞的增殖、EMT 和干性特征。我们的研究表明,PIEZO1 通过激活 Ca2+/Calpain/YAP 通路,在介导 KIRC 生长和转移中发挥了关键作用。 结论:本研究阐明了一种新机制,通过该机制,PIEZO1 的激活可导致钙离子流入、随后的钙蛋白酶激活和 YAP 核转位,从而促进基质僵化驱动的 KIRC 进展。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
10.00
自引率
2.00%
发文量
151
审稿时长
44 days
期刊介绍:
BBA Molecular Cell Research focuses on understanding the mechanisms of cellular processes at the molecular level. These include aspects of cellular signaling, signal transduction, cell cycle, apoptosis, intracellular trafficking, secretory and endocytic pathways, biogenesis of cell organelles, cytoskeletal structures, cellular interactions, cell/tissue differentiation and cellular enzymology. Also included are studies at the interface between Cell Biology and Biophysics which apply for example novel imaging methods for characterizing cellular processes.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


