Electroporation-mediated novel albumin-fused Flt3L DNA delivery promotes cDC1-associated anticancer immunity
IF 4.5
3区 医学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
Dendritic cells (DCs) constitute a distinct type of immune cell found within tumors, serving a central role in mediating tumor antigen-specific immunity against cancer cells. Frequently, DC functions are dysregulated by the immunosuppressive signals present within the tumor microenvironment (TME). Consequently, DC manipulation holds great potential to enhance the cytotoxic T cell response against cancer diseases. One strategy involves administering Fms-like tyrosine kinase receptor 3 ligand (Flt3L), a vitally important cytokine for DC development. In this current study, the electroporation-mediated delivery of a novel albumin-fused Flt3L DNA (alb-Flt3L DNA) demonstrated the ability to induce an anti-tumor immune response. This albumin fusion construct possesses more persistent bioactivity in targeted organs. Furthermore, TC-1-bearing-C57BL/6 mice receiving alb-Flt3L DNA treatment presented better tumor control and superior survival. Cellular analysis revealed that alb-Flt3L DNA administration promoted robust DC and cDC1 expansion. In addition, increased levels of IFN-γ-secreting CD8+ lymphocytes were found in correlation to greater cDC1 population. Moreover, the toxicity of alb-Flt3L administration is limited. Collectively, our data showcases a novel DC-based immunotherapy using electroporation to administer alb-Flt3L DNA.
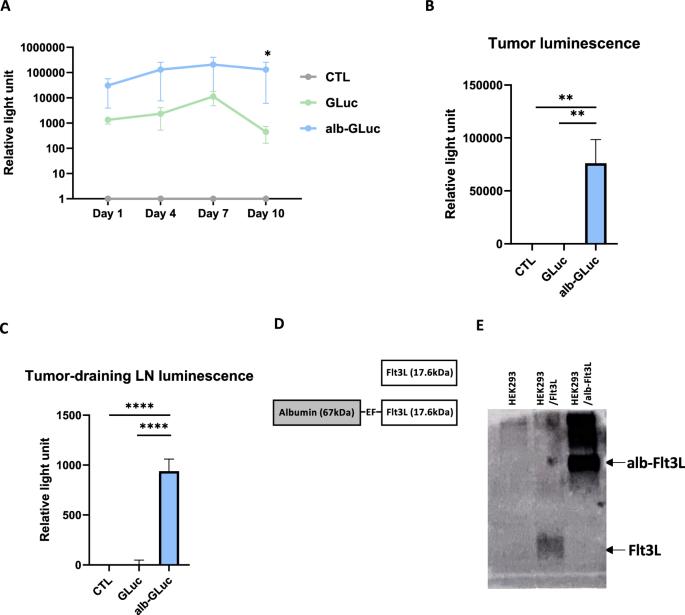
电穿孔介导的新型白蛋白融合 Flt3L DNA 递送可促进 cDC1 相关的抗癌免疫。
树突状细胞(DC)是肿瘤内一种独特的免疫细胞,在介导针对癌细胞的肿瘤抗原特异性免疫中发挥着核心作用。肿瘤微环境(TME)中存在的免疫抑制信号经常会导致树突状细胞功能失调。因此,操纵直流电具有增强细胞毒性 T 细胞对癌症疾病反应的巨大潜力。其中一种策略是施用Fms样酪氨酸激酶受体3配体(Flt3L),这是一种对DC发育至关重要的细胞因子。在目前的研究中,电穿孔介导的新型白蛋白融合 Flt3L DNA(alb-Flt3L DNA)递送证明了诱导抗肿瘤免疫反应的能力。这种白蛋白融合构建体在靶器官中具有更持久的生物活性。此外,接受 alb-Flt3L DNA 治疗的 TC-1-C57BL/6 小鼠的肿瘤控制率更高,存活率更高。细胞分析表明,alb-Flt3L DNA 能促进 DC 和 cDC1 的扩增。此外,分泌 IFN-γ 的 CD8+ 淋巴细胞水平的增加与 cDC1 数量的增加相关。此外,施用 alb-Flt3L 的毒性有限。总之,我们的数据展示了一种新型的基于直流电的免疫疗法,它利用电穿孔技术给药 alb-Flt3L DNA。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Gene Therapy
医学-生化与分子生物学
CiteScore
9.70
自引率
2.00%
发文量
67
审稿时长
4-8 weeks
期刊介绍:
Gene Therapy covers both the research and clinical applications of novel therapeutic techniques based on a genetic component. Over the last few decades, significant advances in technologies ranging from identifying novel genetic targets that cause disease through to clinical studies, which show therapeutic benefit, have elevated this multidisciplinary field to the forefront of modern medicine.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


