Characterization of anti-AAV2 neutralizing antibody levels in sheep prior to and following intravitreal AAV2.7m8 injection
IF 4.5
3区 医学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
Gene augmentation therapy is a promising treatment for incurable, blinding inherited retinal diseases, and intravitreal delivery is being studied as a safe alternative to subretinal injections. Adeno-Associated Viruses (AAV) are commonly-used vectors for ocular gene augmentation therapy. Naturally occurring pre-operative exposure and infection with AAV could result in presence of neutralizing antibodies (NAB’s) in patients’ serum, and may affect the safety and efficacy of treatment. Our aim was to characterize the humoral response against AAV pre- and post-intravitreal delivery of AAV2.7m8 vectors in a naturally-occurring sheep model of CNGA3 achromatopsia. Serial serum neutralization assays were performed to screen sheep for pre-exiting anti-AAV2 NAB’s, and to assess the effect of intravitreal AAV2.7m8 injection on post-operative NAB titers and intraocular inflammation in sheep. The effect of viral dose and transgene type were also assessed. Serological screening revealed pre-operative seropositivity in 21.4% of animals, with age being a risk factor for the presence of anti-AAV2 NAB’s. NAB titers increased following intravitreal AAV administration in the majority of sheep. There was no significant difference in the degree of post-operative serum neutralization between pre-operatively seronegative sheep and those with pre-existing antibodies. However, only sheep with pre-existing antibodies presented with signs of post-operative inflammation. We conclude that pre-existing anti-AAV2 NAB’s do not affect the level of post-operative NAB titers; however, they increase the risk of post-operative ocular inflammation. Our results could have implications for the management of AAV-mediated ocular gene therapies, a technology being increasingly studied and used in patients.
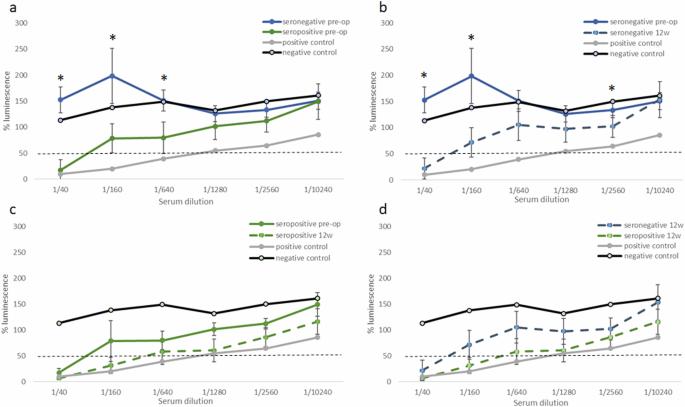
玻璃体内注射 AAV2.7m8 之前和之后绵羊体内抗 AAV2 中和抗体水平的特征。
基因扩增疗法是治疗无法治愈的致盲性遗传性视网膜疾病的一种很有前景的方法,目前正在研究将玻璃体内给药作为视网膜下注射的一种安全替代方法。腺相关病毒(AAV)是眼部基因增强疗法常用的载体。术前自然发生的 AAV 暴露和感染可能导致患者血清中出现中和抗体(NAB),并可能影响治疗的安全性和有效性。我们的目的是在自然发生的 CNGA3 失色素性眼病绵羊模型中,分析 AAV2.7m8 载体静脉注射前后对 AAV 的体液反应。通过连续血清中和试验筛查绵羊体内预先存在的抗 AAV2 NAB,并评估玻璃体内注射 AAV2.7m8 对绵羊术后 NAB 滴度和眼内炎症的影响。此外,还评估了病毒剂量和转基因类型的影响。血清学筛查显示,21.4%的动物术前血清阳性,年龄是出现抗AAV2 NAB的风险因素。大多数绵羊在玻璃体内注射 AAV 后,NAB 滴度都会升高。术前血清阴性的绵羊和已有抗体的绵羊在术后血清中和程度上没有明显差异。然而,只有存在抗体的绵羊才会出现术后炎症症状。我们的结论是,术前存在的抗 AAV2 NAB 不会影响术后 NAB 滴度水平,但会增加术后眼部发炎的风险。我们的研究结果可能会对 AAV 介导的眼部基因疗法的管理产生影响,这项技术正在被越来越多的患者研究和使用。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Gene Therapy
医学-生化与分子生物学
CiteScore
9.70
自引率
2.00%
发文量
67
审稿时长
4-8 weeks
期刊介绍:
Gene Therapy covers both the research and clinical applications of novel therapeutic techniques based on a genetic component. Over the last few decades, significant advances in technologies ranging from identifying novel genetic targets that cause disease through to clinical studies, which show therapeutic benefit, have elevated this multidisciplinary field to the forefront of modern medicine.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


