Thymidine phosphorylase mediates SARS-CoV-2 spike protein enhanced thrombosis in K18-hACE2TG mice
IF 3.7
3区 医学
Q1 HEMATOLOGY
引用次数: 0
Abstract
Introduction
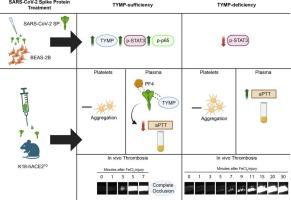
Thymidine phosphorylase (TYMP), which facilitates platelet activation and thrombosis, is significantly increased in COVID-19 patients. We hypothesize that TYMP mediates SARS-CoV-2 spike protein (SP)-induced thrombosis.
Materials and methods
Plasmids encoding wildtype SP or empty vector (p3.1) were transfected into COS-7 cells, and cell lysates were prepared as a reservoir for SP or p3.1 (control), respectively. K18-hACE2TG and K18-hACE2TG/Tymp−/− mice were treated with a single dose of SP or p3.1 by intraperitoneal injection and then subjected to thrombosis studies three days later. The role of SP on inflammatory signaling activation was assessed in BEAS-2B cells.
Results
SARS-CoV-2 SP increased the expression of TYMP, resulting in the activation of STAT3 and NF-κB in BEAS-2B cells. A siRNA-mediated knockdown of TYMP attenuated SP-enhanced activation of STAT3. SP significantly promoted arterial thrombosis in K18-hACE2TG mice. SP-accelerated thrombosis was attenuated by inhibition or genetic ablation of TYMP. SP treatment did not influence ADP- or collagen-induced platelet aggregation but significantly increased platelet adhesion to fibrinogen. SP treatment also significantly shortened activated partial thromboplastin time, which was reversed and even prolonged by TYMP deficiency. Additionally, SP binds to platelet factor 4 (PF4) and TYMP. TYMP does not bind PF4 but enhances the formation of the SP/PF4 complex, which may augment the procoagulant and prothrombotic effect of PF4.
Conclusions
We conclude that SP is prothrombotic and upregulates TYMP expression, and TYMP inhibition or knockout mitigates SP-enhanced thrombosis. These findings suggest that inhibition of TYMP may be a novel therapeutic strategy for COVID-19-associated thrombosis.
胸苷磷酸化酶在 K18-hACE2TG 小鼠体内介导了 SARS-CoV-2 穗状病毒血栓形成的增强。
简介胸苷磷酸化酶(TYMP)可促进血小板活化和血栓形成,在COVID-19患者中显著增加。我们假设胸苷磷酸化酶介导了 SARS-CoV-2 穗状蛋白(SP)诱导的血栓形成:将编码野生型 SP 或空载体(p3.1)的质粒转染到 COS-7 细胞中,并分别制备细胞裂解液作为 SP 或 p3.1 (对照)的储备液。K18-hACE2TG 和 K18-hACE2TG/Tymp-/- 小鼠腹腔注射单剂量 SP 或 p3.1,三天后进行血栓研究。在BEAS-2B细胞中评估了SP对炎症信号激活的作用:结果:SARS-CoV-2 SP增加了TYMP的表达,导致STAT3和NF-κB在BEAS-2B细胞中被激活。siRNA 介导的 TYMP 敲除抑制了 SP 增强的 STAT3 激活。SP能明显促进K18-hACE2TG小鼠动脉血栓的形成。抑制或基因消融 TYMP 可减轻 SP 加速的血栓形成。SP 处理不会影响 ADP 或胶原诱导的血小板聚集,但会显著增加血小板对纤维蛋白原的粘附。SP 处理还能明显缩短活化部分凝血活酶时间,而 TYMP 缺乏会逆转甚至延长该时间。此外,SP 还能与血小板因子 4(PF4)和 TYMP 结合。TYMP 不与 PF4 结合,但能增强 SP/PF4 复合物的形成,这可能会增强 PF4 的促凝血和促血栓形成作用:我们得出结论:SP 有促血栓形成的作用,并能上调 TYMP 的表达,而抑制或敲除 TYMP 能减轻 SP 增强的血栓形成。这些发现表明,抑制TYMP可能是治疗COVID-19相关血栓形成的一种新策略。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Thrombosis research
医学-外周血管病
CiteScore
14.60
自引率
4.00%
发文量
364
审稿时长
31 days
期刊介绍:
Thrombosis Research is an international journal dedicated to the swift dissemination of new information on thrombosis, hemostasis, and vascular biology, aimed at advancing both science and clinical care. The journal publishes peer-reviewed original research, reviews, editorials, opinions, and critiques, covering both basic and clinical studies. Priority is given to research that promises novel approaches in the diagnosis, therapy, prognosis, and prevention of thrombotic and hemorrhagic diseases.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


