Increased hydrogen sulfide turnover serves a cytoprotective role during the development of replicative senescence
IF 5.3
2区 医学
Q1 PHARMACOLOGY & PHARMACY
引用次数: 0
Abstract
The mammalian gasotransmitter hydrogen sulfide (H2S) is produced by enzymes such as cystathionine β-synthase (CBS), cystathionine γ-lyase (CSE), 3-mercaptopyruvate sulfurtransferase (3-MST). Prior studies suggest that H2S may have cytoprotective and anti-aging effects. This project explores the regulation and role of endogenous H2S in a murine model of replicative senescence. H2S and polysulfide levels in RAW 264.7 murine macrophages (control cells: passage 5–10; senescent cells: passage 30–40) were measured using fluorescent probes. The expression of H2S-related enzymes and the activity of senescence marker beta-galactosidase (SA-β-Gal) were also analyzed. CBS, CSE, and 3-MST were inhibited using selective pharmacological inhibitors. Senescence led to a moderate upregulation of CBS and in a significant increase in CSE and 3-MST. H2S degradation enzymes were also elevated in senescence. Inhibition of H2S-producing enzymes reduced H2S levels but increased polysulfides. Inhibition of H2S production during senescence suppressed cell proliferation, and elevated SA-β-Gal and p21 levels. Comparing young and old mice spleens revealed downregulation of CBS and ETHE1 and upregulation of rhodanese and SUOX in older mice. The results demonstrate that increased reactive sulfur turnover occurs in senescent macrophages and that reactive sulfur species support cell proliferation and regulate cellular senescence.
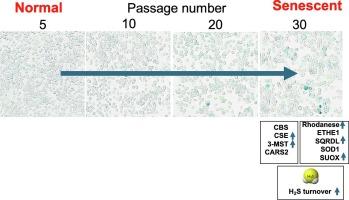
在复制衰老的发展过程中,硫化氢周转的增加起到了细胞保护的作用。
哺乳动物的气体递质硫化氢(H2S)是由胱硫醚β-合成酶(CBS)、胱硫醚γ-赖氨酸酶(CSE)、3-巯基丙酮酸硫基转移酶(3-MST)等酶产生的。先前的研究表明,H2S 可能具有细胞保护和抗衰老作用。本项目探讨了内源性 H2S 在小鼠复制性衰老模型中的调节和作用。使用荧光探针测量了 RAW 264.7 小鼠巨噬细胞(对照细胞:5-10 期;衰老细胞:30-40 期)中的 H2S 和多硫化物水平。此外,还分析了 H2S 相关酶的表达和衰老标记物 beta-半乳糖苷酶(SA-β-Gal)的活性。使用选择性药理抑制剂抑制了 CBS、CSE 和 3-MST。衰老导致 CBS 中度上调,CSE 和 3-MST 显著增加。在衰老过程中,H2S 降解酶也会升高。抑制产生 H2S 的酶会降低 H2S 水平,但会增加多硫化物。抑制衰老期的 H2S 生成可抑制细胞增殖,并提高 SA-β-Gal 和 p21 的水平。比较年轻小鼠和老年小鼠的脾脏发现,老年小鼠的 CBS 和 ETHE1 下调,而红丹和 SUOX 上调。研究结果表明,衰老的巨噬细胞中活性硫周转增加,活性硫支持细胞增殖并调节细胞衰老。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Biochemical pharmacology
医学-药学
CiteScore
10.30
自引率
1.70%
发文量
420
审稿时长
17 days
期刊介绍:
Biochemical Pharmacology publishes original research findings, Commentaries and review articles related to the elucidation of cellular and tissue function(s) at the biochemical and molecular levels, the modification of cellular phenotype(s) by genetic, transcriptional/translational or drug/compound-induced modifications, as well as the pharmacodynamics and pharmacokinetics of xenobiotics and drugs, the latter including both small molecules and biologics.
The journal''s target audience includes scientists engaged in the identification and study of the mechanisms of action of xenobiotics, biologics and drugs and in the drug discovery and development process.
All areas of cellular biology and cellular, tissue/organ and whole animal pharmacology fall within the scope of the journal. Drug classes covered include anti-infectives, anti-inflammatory agents, chemotherapeutics, cardiovascular, endocrinological, immunological, metabolic, neurological and psychiatric drugs, as well as research on drug metabolism and kinetics. While medicinal chemistry is a topic of complimentary interest, manuscripts in this area must contain sufficient biological data to characterize pharmacologically the compounds reported. Submissions describing work focused predominately on chemical synthesis and molecular modeling will not be considered for review.
While particular emphasis is placed on reporting the results of molecular and biochemical studies, research involving the use of tissue and animal models of human pathophysiology and toxicology is of interest to the extent that it helps define drug mechanisms of action, safety and efficacy.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


