Lack of AMP-activated protein kinase-α1 reduces nitric oxide synthesis in thoracic aorta perivascular adipose tissue
IF 3.5
3区 医学
Q2 PHARMACOLOGY & PHARMACY
引用次数: 0
Abstract
Objective
Perivascular adipose tissue (PVAT) releases anti-contractile bioactive molecules including NO. PVAT anti-contractile activity is attenuated in mice lacking AMPKα1 (AMP-activated protein kinase-α1). As AMPK regulates endothelial NO synthase (eNOS) activity in cultured cells, NO synthesis was examined in PVAT from AMPKα1 knockout (KO) mice.
Methods and results
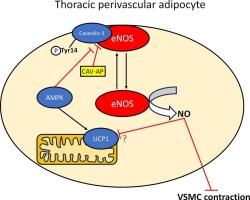
Endothelium-denuded thoracic or abdominal aortic rings were isolated from wild type (WT) and KO mice. NOS inhibition enhanced vasoconstriction in PVAT-intact thoracic aortic rings from mice of either genotype yet had no effect on abdominal rings as assessed by wire myography. Thoracic aorta PVAT exhibited increased NO production, NOS activity and levels of the brown adipose tissue marker uncoupling protein-1 (UCP1) compared to abdominal PVAT. In KO mice, NO production was significantly reduced in thoracic but not abdominal PVAT. Reduced NO production in KO thoracic PVAT was not due to altered levels or phosphorylation of eNOS but was associated with increased caveolin-1:eNOS association and caveolin-1 Tyr14 phosphorylation. A peptide that disrupts eNOS:caveolin-1 association increased NO synthesis and reduced vasoconstriction of PVAT-intact thoracic but not abdominal aortic rings. KO thoracic PVAT also exhibited reduced UCP1 levels.
Conclusions
Murine thoracic aorta PVAT exhibits higher NO synthesis and UCP1 levels than abdominal aortic PVAT. Downregulation of AMPK suppresses NO synthesis which may contribute to the reduced anticontractile activity and reduced brown adipose tissue phenotype of KO thoracic PVAT. The mechanism underlying the effect of AMPK downregulation likely results from increased caveolin-1:eNOS association associated with caveolin-1 Tyr14 phosphorylation.
缺乏 AMP 活化蛋白激酶-α1 会减少胸主动脉血管周围脂肪组织中一氧化氮的合成。
目的血管周围脂肪组织(PVAT)释放包括 NO 在内的抗收缩生物活性分子。缺乏 AMPKα1 (AMP-活化蛋白激酶-α1)的小鼠的血管周围脂肪组织抗收缩活性减弱。由于 AMPK 在培养细胞中调节内皮 NO 合酶(eNOS)的活性,因此研究了 AMPKα1 基因敲除(KO)小鼠 PVAT 的 NO 合成情况:从野生型(WT)和 KO 小鼠体内分离出内皮剥离的胸主动脉环或腹主动脉环。抑制 NOS 可增强两种基因型小鼠与 PVAT 接触的胸主动脉环的血管收缩,但对腹主动脉环没有影响,这是由线性肌电图评估的。与腹主动脉皮下脂肪细胞相比,胸主动脉皮下脂肪细胞的 NO 产量、NOS 活性和棕色脂肪组织标志物解偶联蛋白-1(UCP1)水平均有所增加。在 KO 小鼠中,胸主动脉瓣的 NO 生成明显减少,而腹主动脉瓣则没有。KO 小鼠胸腔腹腔静脉血管内氮氧化物生成的减少不是由于 eNOS 水平或磷酸化的改变,而是与洞穴素-1:eNOS 结合和洞穴素-1 Tyr14 磷酸化的增加有关。一种能破坏 eNOS:caveolin-1 关联的多肽能增加 NO 的合成,并减少与 PVAT 接触的胸主动脉环的血管收缩,但不能减少腹主动脉环的血管收缩。KO 的胸主动脉瓣也显示出 UCP1 水平降低:结论:与腹主动脉 PVAT 相比,小鼠胸主动脉 PVAT 的 NO 合成和 UCP1 水平更高。AMPK的下调抑制了NO的合成,这可能是KO胸主动脉瓣抗收缩活性降低和棕色脂肪组织表型减少的原因。AMPK 下调的影响机制可能是与洞穴素-1 Tyr14 磷酸化相关的洞穴素-1:eNOS 关联增加所致。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Vascular pharmacology
医学-药学
CiteScore
6.60
自引率
2.50%
发文量
153
审稿时长
31 days
期刊介绍:
Vascular Pharmacology publishes papers, which contains results of all aspects of biology and pharmacology of the vascular system.
Papers are encouraged in basic, translational and clinical aspects of Vascular Biology and Pharmacology, utilizing approaches ranging from molecular biology to integrative physiology. All papers are in English.
The Journal publishes review articles which include vascular aspects of thrombosis, inflammation, cell signalling, atherosclerosis, and lipid metabolism.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


