Common structural features in some of the sequentially distant neurotransmitter transporters N-termini
IF 2.7
3区 生物学
Q3 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
The N-terminal regions of SLC6 transporters are sequentially unrelated, and the majority of such transporters contain only relatively short peptide N-terminal extensions. Currently, it is not clear if a diversity of N-terminal sequences represents diverse functions among the transporters or if there are common functions hidden behind similar, as yet unidentified, structures. Using alignment of amino acid sequences with the hydropathy plot, disorder prediction, and calpain recognition sites, we show that common structural features among the N-termini of some transporters might exist. We previously showed that polymeric neurotransmitter transporter N-termini exhibit very similar profiles of dynamic, time-dependent 465-595-350-750 nm absorbance metachromasia in the Bradford assay. Here we report that under certain mild denaturing conditions, filamentous aggregation of glutathione S-transferase (GST) protein results in similar near-infrared metachromasia. This effect was eliminated by further GST protein denaturation and solubilization. The results suggest that aggregation of partially denatured GST stabilizes Coomassie dye docking sites, producing a near-infrared absorbance shift similar to that observed in the polymeric unstructured N-termini of transporters.
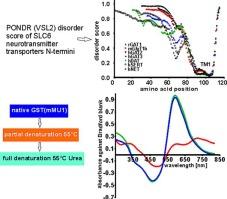
一些相距较远的神经递质转运体 N 端的共同结构特征。
SLC6 转运体的 N 端区域在序列上互不关联,大多数此类转运体只包含相对较短的肽 N 端延伸。目前还不清楚 N 端序列的多样性是代表了转运体的不同功能,还是在相似但尚未确定的结构背后隐藏着共同的功能。我们以前曾发现,在布拉德福德测定法中,聚合神经递质转运体 N 端表现出非常相似的动态、随时间变化的 465 nm-595 nm-750 nm 吸光度变色曲线。我们在此报告,在某些轻度变性条件下,谷胱甘肽 S-转移酶(GST)蛋白的丝状聚集会导致类似的近红外变色。进一步变性和增溶谷胱甘肽 S 转移酶蛋白后,这种效应就会消失。研究结果表明,部分变性的 GST 聚合会稳定库马西染料对接位点,产生类似于在转运体聚合非结构 N 端观察到的近红外吸光度偏移。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of structural biology
生物-生化与分子生物学
CiteScore
6.30
自引率
3.30%
发文量
88
审稿时长
65 days
期刊介绍:
Journal of Structural Biology (JSB) has an open access mirror journal, the Journal of Structural Biology: X (JSBX), sharing the same aims and scope, editorial team, submission system and rigorous peer review. Since both journals share the same editorial system, you may submit your manuscript via either journal homepage. You will be prompted during submission (and revision) to choose in which to publish your article. The editors and reviewers are not aware of the choice you made until the article has been published online. JSB and JSBX publish papers dealing with the structural analysis of living material at every level of organization by all methods that lead to an understanding of biological function in terms of molecular and supermolecular structure.
Techniques covered include:
• Light microscopy including confocal microscopy
• All types of electron microscopy
• X-ray diffraction
• Nuclear magnetic resonance
• Scanning force microscopy, scanning probe microscopy, and tunneling microscopy
• Digital image processing
• Computational insights into structure
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


